Involuntary movements antipsychotics
Recognizing Antipsychotic-Induced Movement Disorders
US Pharm. 2015;40(11):22-25.
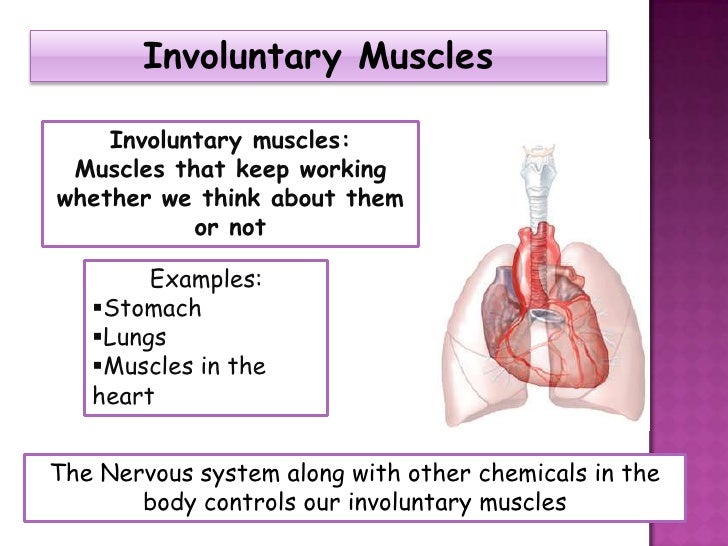
Second-generation antipsychotic agents (SGAs, TABLE 1), also referred to as atypical antipsychotics, are widely used in the United States in the treatment of schizophrenia, psychotic episodes, and other conditions. These agents, as compared with first-generation antipsychotics (FGAs), are associated with a dramatic decline in drug-induced movement disorders (DIMDs) such as akathisia (subjective motor restlessness), dystonia (dyskinetic muscle tonicity), parkinsonism, and tardive dyskinesia (TD; TABLE 2).1
However, the use of SGAs for populations that were antipsychotic agent-naïve, (i.e., elderly, adolescents, children) and an ever-widening adult population with conditions such as bipolar disorder and major depressive disorder, together with a lack of long-term clinical trials that would precisely indicate the rate of DIMD in these populations, portends an increase in the actual number of cases. 2
In addition, the literature on identifiable risk factors associated with treatment-emergent movement disorders is highly variable.3 Recognition of DIMDs is essential in the treatment of these disorders.3 Pharmacists can play a role not only in identifying cases of DIMDs, but importantly, in guiding appropriate SGA dosing in light of their continued widening use in vulnerable populations such as the elderly, in the hope of reducing the incidence of, or preventing, these disorders.2
With the recent FDA approval of the newest SGA, cariprazine (Vraylar) capsules to treat schizophrenia and bipolar disorder in adults, reviewing the issues related to DIMDs is timely indeed. The development of a DIMD often reduces the quality of life in a geriatric patient.3 In particular, elderly patients with dementia are at greater risk than patients without dementia for persistent, drug-induced extrapyramidal symptoms (EPS). Sedation, orthostatic hypotension, anticholinergic effects, and akathisia may occur in up to 20% of elderly patients taking an antipsychotic agent, and drug-induced parkinsonism can persist for 6 to 9 months after the agent is discontinued. 4 Further, tens of thousands of patients previously exposed to more potent antipsychotic agents have TD, which remains untreatable and irreversible.1 Of additional concern, the medications used in the management of DIMDs, including anticholinergic (antiparkinsonism) agents, benzodiazepines, and beta-blockers, require careful consideration in this vulnerable population.1,5
4 Further, tens of thousands of patients previously exposed to more potent antipsychotic agents have TD, which remains untreatable and irreversible.1 Of additional concern, the medications used in the management of DIMDs, including anticholinergic (antiparkinsonism) agents, benzodiazepines, and beta-blockers, require careful consideration in this vulnerable population.1,5
FGAs and SGAs: Focus on Mechanism of Action
In general, antipsychotic drugs block at dopaminergic and serotonergic receptors as well as at adrenergic, cholinergic, and histamine-binding neuroreceptors.6 FGAs are more likely associated with movement disorders, particularly for drugs that bind tightly to dopamine receptors (D2), such as haloperidol, and less likely for those agents that bind weakly, such as chlorpromazine. 7 The SGAs block dopamine receptors more selectively than conventional FGAs.7
According to Caroff, the dominance in clinical practice of the newer SGAs has significantly reduced the frequency of EPS, whether by a reduced liability for EPS because of weaker dopamine receptor–binding affinity or a more complex “atypical” mechanism affecting multiple neurotransmitters. 1 According to Schulz, although greater serotonergic binding was initially thought to contribute to the efficacy of SGAs, studies suggest this binding is unrelated to efficacy or adverse-effects profile; SGAs also do the following7:
1 According to Schulz, although greater serotonergic binding was initially thought to contribute to the efficacy of SGAs, studies suggest this binding is unrelated to efficacy or adverse-effects profile; SGAs also do the following7:
• Have the tendency to alleviate positive symptoms (e.g., delusions, hallucinations)
• Potentially lessen negative symptoms to a greater extent than do FGAs (although clinicians may question this difference)
• Potentially cause less cognitive blunting
• Are less likely to have extrapyramidal adverse effects
• Are associated with a lower risk of TD
• Increase prolactin slightly or not at all (except risperidone)
Of note, while SGAs have fewer EPS than FGAs (TABLE 1), SGAs are associated with a higher risk of metabolic side effects, such as diabetes, hyperlipidemia, and weight gain.6,7
The Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) Schizophrenia Trial provided additional perspective on the relative risks and clinical value of the SGAs and FGAs; the study showed that perphenazine, a mid-potency FGA used at modest doses, was not significantly different from four SGAs in the risk of developing EPS. 1 Caroff indicates that the dichotomy between FGAs and SGAs and the “atypical” concept based on liability of EPS may be oversimplified; instead, he suggests “antipsychotic drugs could be conceptualized as a single drug class with a spectrum of risk for EPS depending upon receptor-binding affinities, especially for dopamine and acetylcholine receptors, and individual patient susceptibility.”1
1 Caroff indicates that the dichotomy between FGAs and SGAs and the “atypical” concept based on liability of EPS may be oversimplified; instead, he suggests “antipsychotic drugs could be conceptualized as a single drug class with a spectrum of risk for EPS depending upon receptor-binding affinities, especially for dopamine and acetylcholine receptors, and individual patient susceptibility.”1
Schizophrenia and Antipsychotics in the Elderly
The average age at onset of schizophrenia is early to mid-20s in women and somewhat earlier in men; about 40% of males have their first episode before age 20.7 With regard to the elderly, late-life onset schizophrenia, sometimes referred to as paraphrenia, may occur.7 Most geriatric patients treated for schizophrenia, however, have been living with the symptoms for decades, including psychosis (loss of contact with reality), hallucinations (false perceptions), delusions (false beliefs), disorganized speech and behavior, flattened affect (restricted range of emotions), cognitive deficits (impaired reasoning and problem solving), and occupational and social dysfunction. 7
7
Furthermore, large numbers of geriatric patients previously treated with haloperidol and other FGAs have TD; these agents are still used in psychiatry; in medical settings for delirium and agitation; and in developing nations.1
While all antipsychotic agents can reduce paranoia, they have the potential to worsen confusion; elderly patients, especially women, are at increased risk of TD, which is often irreversible.4 Since antipsychotics are considered a drug category of concern in the elderly, according to Ruscin and Linnebur, antipsychotics should be used only for psychosis; in nonpsychotic, agitated patients, they indicate that antipsychotics control symptoms only marginally better than placebo and can have severe adverse effects.4
In people with dementia, studies have shown antipsychotics increased mortality and risk of stroke, moving the FDA to issue a black box warning on their use in this population; in general, dementia-related behavior problems, such as wandering, yelling, and uncooperativeness, do not respond to antipsychotics.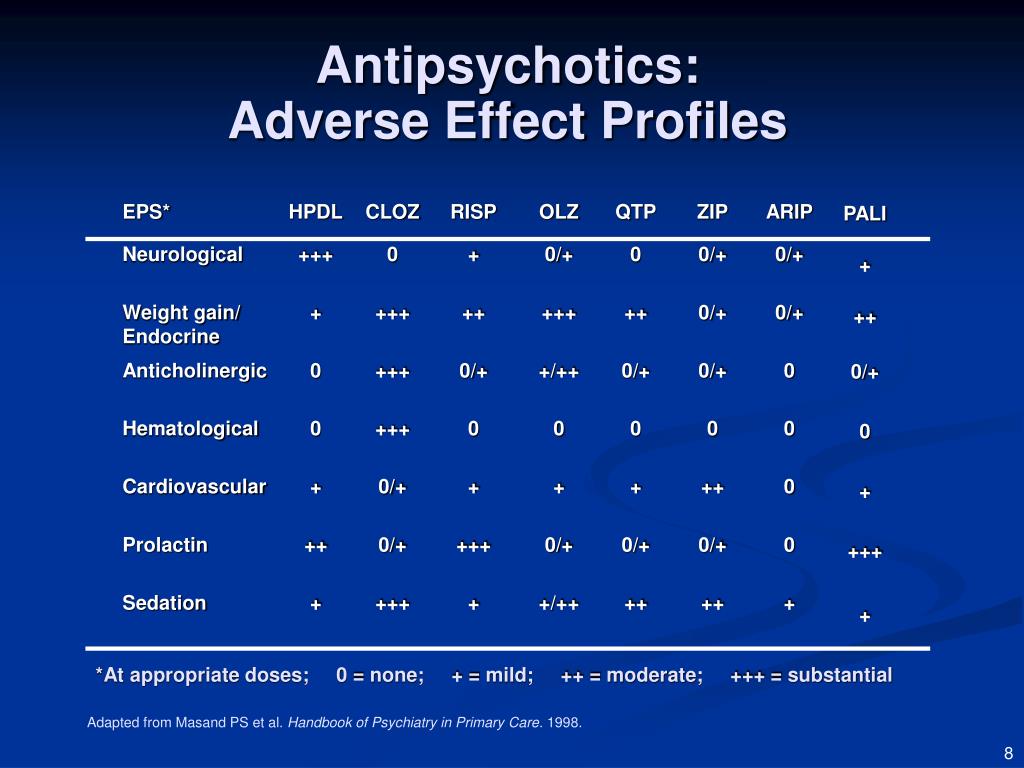 4
4
Regarding further caution in the elderly, antipsychotics may exacerbate or cause syndrome of inappropriate antidiuretic hormone secretion or hyponatremia; it is recommended that sodium levels be closely monitored when starting or changing dosages in older adults.8 When an antipsychotic agent is used with two or more CNS-active drugs (antipsychotics; benzodiazepines; nonbenzodiazepines, benzodiazepine receptor agonist hypnotics; tricyclic antidepressants, selective serotonin reuptake inhibitors, and opioids), there is an increased risk of falls; avoid a total of three or more CNS-active drugs and minimize the number of CNS-active drugs.8
SGA Dosing in the Geriatric Patient
When initiating an antipsychotic agent in an elderly person, the initial dose should be about one- quarter of the usual starting adult dose; it should then be increased gradually with frequent monitoring for response and adverse effects.4 Upon clinical response by the patient, the dose should be decreased, if possible, to the lowest effective dose; it should be discontinued if it is ineffective. Clinical trial data are limited with regard to dosing, efficacy, and safety of these drugs in the elderly.4 As previously mentioned, elderly patients with dementia are at greater risk than patients without dementia for persistent drug-induced EPS.1
Clinical trial data are limited with regard to dosing, efficacy, and safety of these drugs in the elderly.4 As previously mentioned, elderly patients with dementia are at greater risk than patients without dementia for persistent drug-induced EPS.1
Drug-Induced Movement Disorders: EPS
Management requires cooperation from the patient, family, and other caregivers that can be fostered early in the patient’s care through appropriate informed consent. According to Caligiuri, the risks and benefits of antipsychotic treatment in the elderly patient need to be communicated to the patient and the family.3 Once TD develops, there is at present no consistently effective treatment; therefore, attention should focus on its prevention and close monitoring. The pharmacological management of DIMDs requires careful consideration of the precautions and warnings associated with interventions such as anticholinergics, beta-blockers, and benzodiazepines. Refer to the 2015 American Geriatrics Society Updated Beers Criteria for Potentially Inappropriate Medication Use in Older Adults for guidance. 8 DIMDs include:
8 DIMDs include:
Akathisia (i.e., motor restlessness), the most common extrapyramidal symptom, is often mistaken for anxiety or exacerbation of psychosis, and remains a problem even with newer SGA drugs.1,5 The desire to be in motion and the subsequent inability to sit or stand still, resulting in pacing, is so distressing and frightening to patients that it may induce suicidality.5 As indicated in a representative sample of trials, akathisia developed at an incidence rate of about two to seven times higher with haloperidol (15% to 40%) when compared with SGAs (0% to 12%).1 Of note, cigarette smoking in women has been associated with increased incidence.5
While risk factors for akathisia have not been well established, the following may entail greater risk: increasing age, female gender, negative symptoms, cognitive dysfunction, iron deficiency, history of akathisia, concomitant parkinsonism, and mood disorders. 9,10 In most clinical trials, SGAs have been shown to have a significantly lower incidence of akathisia compared with FGAs.11
9,10 In most clinical trials, SGAs have been shown to have a significantly lower incidence of akathisia compared with FGAs.11
While this EPS usually occurs early in the course of antipsychotic treatment, it may persist even after the offending agents are discontinued. While anticholinergic agents (e.g., trihexyphenidyl 2-5 mg orally three time daily) are used to treat akathisia, a benzodiazepine (e.g., clonazepam 0.5-1 mg orally three times daily) is often considered first-line treatment; propranolol 30-80 mg orally/d, diazepam 5 mg orally three times daily, or amantadine 100 mg orally three times daily may be required in resistant cases.5,7 Refer to Reference 19 for geriatric dosing considerations.
Acute dystonias, sustained contractions of muscles leading to twisting, distorted postures, usually occur early in the course of antipsychotic therapy, with younger patients bearing the highest risk.5,6 Late occurrence is seen in individuals who had previously suffered early severe dystonic reactions and an affective disorder, which underscores the importance of thorough medical and medication history-taking in the development of pharmacotherapy care plans.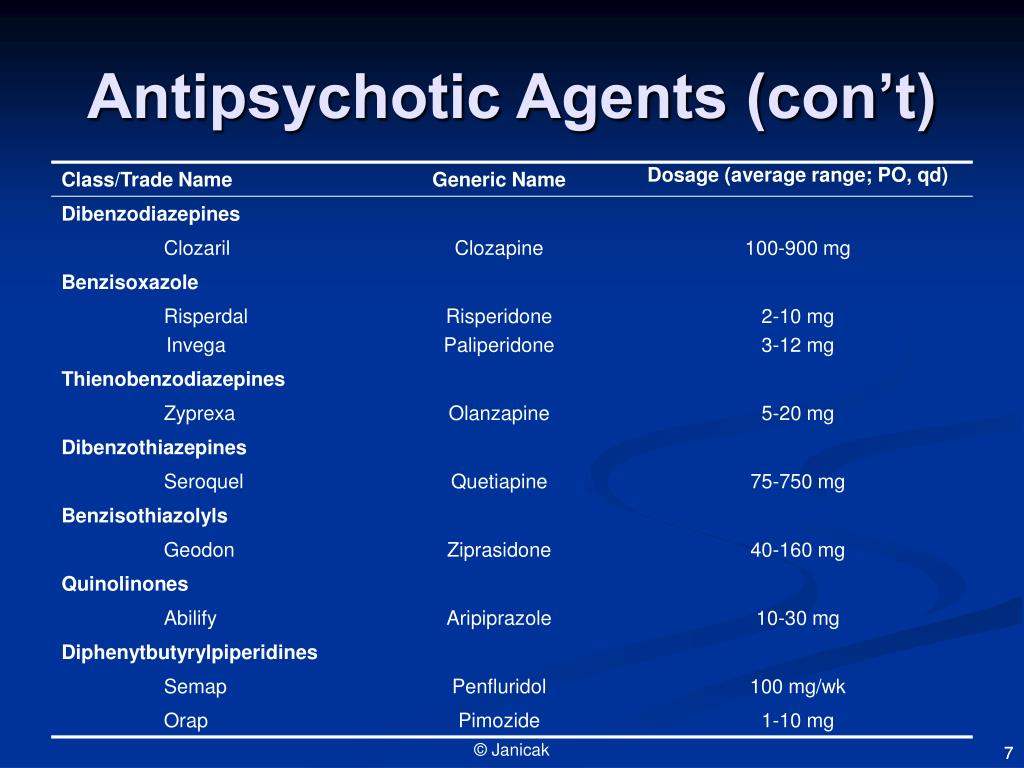 Bizarre muscle spasms of the head, neck, and tongue are most common: torticollis, oculogyric crisis, difficulties in chewing and swallowing, and spasms of the masseter muscle. Particularly dangerous is spasmodic closure of the larynx. While dystonia is less frequent than in years past, it may be encountered with high-potency antipsychotic agents (e.g., haloperidol).1 For acute episodes, diphenhydramine 50 mg is administered intramuscularly; this is followed by the mildly sedating benztropine mesylate, 2 mg orally twice daily, for several weeks, followed by gradual tapering.5
Bizarre muscle spasms of the head, neck, and tongue are most common: torticollis, oculogyric crisis, difficulties in chewing and swallowing, and spasms of the masseter muscle. Particularly dangerous is spasmodic closure of the larynx. While dystonia is less frequent than in years past, it may be encountered with high-potency antipsychotic agents (e.g., haloperidol).1 For acute episodes, diphenhydramine 50 mg is administered intramuscularly; this is followed by the mildly sedating benztropine mesylate, 2 mg orally twice daily, for several weeks, followed by gradual tapering.5
Parkinsonism secondary to drug therapy is reversible, although not distinguishable from idiopathic parkinsonism. Parkinsonism is quite debilitating and more likely to be seen when antipsychotics are used in elderly patients.1 Characteristically, patients present with apathy, akinesia of the face and arms (i.e., appearing as depression), rigidity, salivation, postural abnormalities, shuffling gait, and pill-rolling tremor of the hands; catatonia may also present. 5 Symptoms frequently can be eliminated with a reduction in dose or change to a lower-potency antipsychotic; an anticholinergic, while helpful for drooling, can cause confusion and delirium; however, one may be added if necessary.12 While maximum therapeutic response is usually seen in 3 to 10 days, more severe symptoms may take longer (e.g., 4-6 weeks) to respond. In the elderly, anticholinergics are not for use as prophylaxis against EPS. Amantadine use requires dosing based on renal function. Quetiapine may be a useful SGA alternative for this population.12
5 Symptoms frequently can be eliminated with a reduction in dose or change to a lower-potency antipsychotic; an anticholinergic, while helpful for drooling, can cause confusion and delirium; however, one may be added if necessary.12 While maximum therapeutic response is usually seen in 3 to 10 days, more severe symptoms may take longer (e.g., 4-6 weeks) to respond. In the elderly, anticholinergics are not for use as prophylaxis against EPS. Amantadine use requires dosing based on renal function. Quetiapine may be a useful SGA alternative for this population.12
Tardive dyskinesia (TABLE 2), is more common in female patients, and age is the most consistent risk factor.6,7 Other risk factors, including previous brain injury and alcoholism, are outlined in TABLE 3. TD may be seen in up to 30% of patients receiving long-term FGAs.13-15
While TD is most often mild, it is irreversible, with limited options for treatment. 1 Due to this risk of TD, patients receiving long-term maintenance therapy should be evaluated at least every 6 months; rating instruments may be used.7
1 Due to this risk of TD, patients receiving long-term maintenance therapy should be evaluated at least every 6 months; rating instruments may be used.7
While beyond the scope of this topic, a proposed treatment algorithm for TD may be found in Reference 1.
Neuroleptic malignant syndrome (NMS) is an abrupt, life-threatening, idiosyncratic patient response to a therapeutic dose of an antipsychotic agent; intramuscular administration is a risk factor.6 NMS represents an extremely rare form of EPS combining features of advanced parkinsonism and catatonia; symptoms include exhaustion, agitation, hyperthermia (>38ºC), mental status change, muscle rigidity and other movement disorders, autonomic dysregulation, and elevated creatine kinase (CK).1,7 Lithium, in combination with an antipsychotic, may increase vulnerability.5 NMS requires control of fever and fluid support. The following have been used as treatment, although controversy still exists regarding them: bromocriptine, amantadine, dantrolene, calcium channel blockers, and benzodiazepines; ECT is used in resistant cases, while clozapine is used as an SGA for patients with a history of NMS. 5 Patients and caregivers should be counseled about this adverse reaction and made aware that it requires emergency medical attention.
5 Patients and caregivers should be counseled about this adverse reaction and made aware that it requires emergency medical attention.
Conclusion
The development of a DIMD often reduces the quality of life in a geriatric patient. Pharmacists have an opportunity to implement recommendations to appropriately individualize selection and dosing of an antipsychotic agent, monitor and reassess benefit and risk periodically, and recommend deprescribing where appropriate. Those who are antipsychotic-naïve and especially those who are vulnerable with regard to risk, such as the elderly, often require particular attention.
REFERENCES
1. Caroff SN, Hurford I, Lybrand J, et al. Movement disorders induced by antipsychotic drugs: implications of the CATIE schizophrenia trial. Neurol Clin. 2011; 29(1):127-viii. www.ncbi.nlm.nih.gov/pmc/articles/PMC3018852.
2. Factor SA, Leffler JB, Murray CF. Drug-induced movement disorders: a clinical review. Medscape, 2009. www.medscape.org/viewarticle/586881.Accessed September 22, 2015.
Medscape, 2009. www.medscape.org/viewarticle/586881.Accessed September 22, 2015.
3. Caligiuri MR, Jeste DV, Lacro JP. Antipsychotic-induced movement disorders in the elderly: epidemiology and treatment recommendations. Drugs Aging. 2000;17(5):363-384.
4. Ruscin M, Linnebur SA. Drug categories of concern in the elderly. Merckmanuals.com. Last full review/revision June 2014. www.merckmanuals.com/professional/geriatrics/drug-therapy-in-the-elderly/drug-categories-of-concern-in-the-elderly. Accessed October 27, 2015.
5. Williams N, DeBattista C. Psychiatric disorders. In: Papadakis MA, McPhee SJ, Rabow MW, eds. 2016 Current Medical Diagnosis & Treatment. 55th ed. New York, NY: McGraw Hill Medical; 2015:1050-1056.
6. Harvey RA. Pharmacology. 5th ed. Lippincott Williams & Wilkins: Philadelphia, PA. 2012:161-168.
7. Schulz, C. Schizophrenia. Merckmanuals.com. Last full review/revision July 2013. www.merckmanuals.com/professional/psychiatric-disorders/schizophrenia-and-related-disorders/schizophrenia. Accessed October 27, 2015.
Accessed October 27, 2015.
8. American Geriatrics Society 2015 Updated Beers Criteria for potentially inappropriate medication use in older adults. American Geriatrics Society 2015 Beers Criteria Update Expert Panel. Published Oct 8, 2015. http://onlinelibrary.wiley.com/doi/10.1111/jgs.13702/full. Accessed October 27, 2015.
9. Bratti IM, Kane JM, Marder SR. Chronic restlessness with antipsychotics. Am J Psychiatry. 2007;164(11):1648-1654.
10. Poyurovsky M. Acute antipsychotic-induced akathisia revisited. Br J Psychiatry. 2010;196(2):89-91.
11. Kane JM, Fleischhacker WW, Hansen L, et al. Akathisia: an updated review focusing on second-generation antipsychotics. J Clin Psychiatry. 2009;70(5):627-643.
12. Mathews M, Gratz S, George V, et al. Antipsychotic-induced movement disorders: evaluation and treatment. Psychiatry (Edgmont). 2005;2(3):36-41.
13. Jeste DV, Caliguiri MP. Tardive dyskinesia. Schizophrenia Bull. 1993;19:303-315.
14. Wojcik JD, Gelenberg A, LaBrie RA, et al. Prevalence of TD in an out-patient population. Compr Psychiatry. 1980;21:370-380.
Wojcik JD, Gelenberg A, LaBrie RA, et al. Prevalence of TD in an out-patient population. Compr Psychiatry. 1980;21:370-380.
15. Barnes TRE. Movement disorders associated with antipsychotic drugs: the tardive syndromes. Int Rev Psychiatry. 1990;2:355-366.
16. Woods SW. Chlorpromazine equivalent doses for the newer atypical antipsychotics. J Clin Psychiatry. 2003;64(6):663-667.
17. Vraylar Full Prescribing Information. Actavis Pharma, Inc. Parsippany, NJ. 2015. http://pi.actavis.com/data_stream.asp?product_group=2028&p=pi&language=E. Accessed October 19, 2015.
18. Rexulti Full Prescribing Information. Otsuka Pharmaceutical Co., Ltd., Tokyo, 101-8535 Japan. July 2015. www.rexulti.com. Accessed September 18, 2015.
19. Semla TP, Beizer JL, Higbee MD. Geriatric Dosage Handbook. 19th ed. Hudson, OH: Lexicomp; 2014.
To comment on this article, contact [email protected].
6 Things to Know About Tardive Dyskinesia
Tardive dyskinesia, a drug-induced movement disorder that can cause involuntary lip puckering, tongue movements, frowning, and more, can be a side effect of long-term use of antipsychotic drugs, typically those prescribed for conditions such as schizophrenia, schizoaffective disorder, and bipolar disorder.
Antipsychotics work in part by blocking dopamine receptors in your brain, but sometimes they have unintended consequences for sensory and motor function.
“By definition, tardive dyskinesia is an iatrogenic disorder, meaning it is due to medications,” says Joseph Jankovic, MD, a professor of neurology and the director of the Parkinson’s disease center and movement disorders clinic at Baylor College of Medicine in Houston. “It doesn’t occur without exposure to these medications.”
About 25 percent of people who use first-generation or second-generation antipsychotics long term develop tardive dyskinesia, according to a meta-analysis published in the March 2017 issue of the Journal of Clinical Psychiatry. A third generation of antipsychotics is now available, but some risk remains, says Dr. Jankovic.
Before you take any new medication for the conditions listed above, therefore, have a thorough discussion with your doctor about the risks and benefits.
Here’s what you should know about tardive dyskinesia.
1. Symptoms Often Include Involuntary Mouth Movements
Tardive dyskinesia symptoms can include involuntary mouth movements that resemble chewing, puckering, smacking, or pursing the lips, which can interfere with your ability to speak or chew. Other common symptoms include rapid eye blinking and tongue thrusting.
Tardive dyskinesia can also have sensory symptoms, such as a burning sensation in the mouth, and some women experience vaginal burning, says Jankovic. Young people with tardive dyskinesia may experience tardive dystonia — muscle spasms that may cause involuntary head or neck movements, he says.
2. Symptoms May Not Be Obvious at First
“The onset of tardive dyskinesia is often very subtle,” says Jankovic. “It may initially be manifested by just a feeling of restlessness or the need to move the lips, jaw, or tongue.”
But some people have an immediate reaction to antipsychotic drugs, such as sudden, involuntary, or painful jaw or eye movements.
“That may last a few minutes and may spontaneously resolve,” says Jankovic. “But that often is a signal that there’s more trouble to come. If the person is exposed again to these drugs in the future, it may evolve into this persistent, possibly permanent tardive dyskinesia.”
3. Don’t Stop Taking Antipsychotic Medications Without Your Doctor’s Supervision
If you suspect that you have tardive dyskinesia, consult the healthcare provider who prescribed your antipsychotic medication, says Jankovic.
According to a study published in the May 2018 issue of the Journal of Clinical Psychiatry, doctors can administer the Abnormal Involuntary Movement Scale (AIMS) exam, a short screening for tardive dyskinesia. During this simple test, your doctor will ask a few questions and observe you while you make a few movements.
If it’s determined that you have tardive dyskinesia, your doctor will help you develop a plan, which may include tapering your dose of the medication that's causing it.
“The drug should not be stopped suddenly by the patient,” says Jankovic. “It should be done under the supervision of the physician and should be done gradually.”
4. Drugs Called VMAT2 Inhibitors May Help Relieve Tardive Dyskinesia
Stopping the medication — under your doctor’s supervision only! — is typically the first step to curbing the symptoms of tardive dyskinesia, says Jankovic.
A type of medication called a vesicular monoamine transporter-2 inhibitor, or VMAT2 inhibitor, can also be prescribed to suppress the involuntary movements. Two such drugs, Austedo (deutetrabenazine) and Ingrezza (valbenazine), are approved by the U.S. Food and Drug Administration (FDA) for the treatment of tardive dyskinesia.
5. Tardive Dyskinesia Can Take Months or Years to Subside
Your prognosis with tardive dyskinesia depends on several factors. For one, people who have taken higher doses of antipsychotics or have taken them for longer periods tend to have longer-lasting symptoms.
“Almost all patients with tardive dyskinesia have the condition for months or years, and, particularly in elderly women, it may be a persistent, permanent condition,” says Jankovic. “The sooner they bring symptoms to the attention of the physician and gradually discontinue the medication, the greater likelihood that the condition will resolve.”
People with certain health problems, such as diabetes or a prior brain injury, may also have a harder time finding relief from tardive dyskinesia.
6. Some People Are More Susceptible to Tardive Dyskinesia Than Others
People with certain risk factors are more likely to develop tardive dyskinesia when taking antipsychotics. According to a review published in the June 2018 issue of the Journal of the Neurological Sciences, those risk factors include:
- Being elderly
- Being female
- An intellectual disability or brain damage
- Negative symptoms in schizophrenia
- Mood disorders or cognitive symptoms in mood disorders
- Certain genetic risk factors
- Taking antipsychotics for longer durations
- Diabetes
- Smoking
- Abuse of alcohol or other substances
- Taking first-generation antipsychotics
- Higher doses of an antipsychotic
- Having early parkinsonian side effects
- Taking anticholinergic drugs in addition to antipsychotic medications
- Certain other movement disorders
6 Things You Need to Stop Saying to People With MS — and Why
By Jennifer SchroppWhy I Focus on Today, Not Yesterday
By Trevis Gleason9 Top Remedies for Menstrual Migraine
Get an expert’s advice on how best to prevent menstrual and menstrually related migraine.
By Becky Upham
What Is Neuromyelitis Optica Spectrum Disorder (NMOSD)? Symptoms, Causes, Diagnosis, Treatment, and Prevention
By Quinn PhillipsMS Lesions: How Much Does Location Matter?
By Trevis GleasonWhat You Need to Know About the COVID-19 Vaccine if You Have MS
Experts recommend that most people with multiple sclerosis, as well as members of their household, get the COVID-19 vaccine — but there are a few key ...
By Brian P. Dunleavy
What Is Tardive Dyskinesia? Symptoms, Causes, Diagnosis, Treatment, and Prevention
By Paula DerrowFGBNU NTsPZ.
 ‹‹General Psychiatry››
‹‹General Psychiatry›› Side effects and complications in the treatment of psychotropic drugs
Side effects in psychopharmacotherapy, as in the use of many other drugs, are associated with the inability to selectively influence only pathologically altered brain systems. Some of them are directly related to the therapeutic effect of drugs and occur in most patients taking this drug. As an example, neuroleptic syndrome can be given when using first-generation antipsychotics. Other side effects and complications, which are usually rare, are due to individual patient reactions to a particular drug. In this section, only the most typical side effects and complications associated with the use of psychopharmacological agents of various classes will be considered.
Antipsychotics. The main side effects in the treatment of neuroleptics form neuroleptic syndrome. The leading clinical manifestations of this syndrome are extrapyramidal disorders with a predominance of either hypo- or hyperkinetic disorders. Hypokinetic disorders include drug-induced parkinsonism, manifested by increased muscle tone, lockjaw, rigidity, stiffness, and slowness of movement and speech. Hyperkinetic disorders include tremor, hyperkinesis (choreiform, athetoid, etc.). Usually in the clinical picture in various ratios there are both hypo- and hyperkinetic disorders. The phenomena of dyskinesia may be paroxysmal in nature. Most often they are localized in the mouth area and are manifested by spasmodic contractions of the muscles of the pharynx, tongue, lips, jaws, but often spread to other muscle groups (oculogiric crises, torticollis, torsion spasm, excitomotor crises). Along with extrapyramidal disorders, akathisia phenomena can be observed - feelings of restlessness, "restlessness in the legs", combined with tasikinesia (the need to move, change position). In severe cases, akathisia is accompanied by anxiety, agitation, sleep disorders. A special group of dyskinesia includes tardive dyskinesia (tardive dyskinesia), expressed in involuntary movements of the lips, tongue, face, less often - choreiform movements of the limbs.
Hypokinetic disorders include drug-induced parkinsonism, manifested by increased muscle tone, lockjaw, rigidity, stiffness, and slowness of movement and speech. Hyperkinetic disorders include tremor, hyperkinesis (choreiform, athetoid, etc.). Usually in the clinical picture in various ratios there are both hypo- and hyperkinetic disorders. The phenomena of dyskinesia may be paroxysmal in nature. Most often they are localized in the mouth area and are manifested by spasmodic contractions of the muscles of the pharynx, tongue, lips, jaws, but often spread to other muscle groups (oculogiric crises, torticollis, torsion spasm, excitomotor crises). Along with extrapyramidal disorders, akathisia phenomena can be observed - feelings of restlessness, "restlessness in the legs", combined with tasikinesia (the need to move, change position). In severe cases, akathisia is accompanied by anxiety, agitation, sleep disorders. A special group of dyskinesia includes tardive dyskinesia (tardive dyskinesia), expressed in involuntary movements of the lips, tongue, face, less often - choreiform movements of the limbs. The very name "tardive dyskinesia" suggests that it occurs after long-term treatment with antipsychotics (on average after 2 years). In these cases, there is no correlation with the type of drug, doses and characteristics of treatment at earlier stages, including with previous extrapyramidal disorders.
The very name "tardive dyskinesia" suggests that it occurs after long-term treatment with antipsychotics (on average after 2 years). In these cases, there is no correlation with the type of drug, doses and characteristics of treatment at earlier stages, including with previous extrapyramidal disorders.
Among the disorders of the autonomic nervous system, orthostatic hypotension is most often observed (it is not recommended to stop it with adrenaline), sweating, weight gain, changes in appetite, constipation, diarrhea. Sometimes there are anticholinergic effects - visual disturbances, dysuric phenomena. Possible functional disorders of the cardiovascular system with changes in the ECG in the form of an increase in the interval Q - T , reduction of the gili tooth or its inversion, tachy or bradycardia. Sometimes there are side effects in the form of photosensitivity, dermatitis, skin pigmentation; skin allergic reactions are possible. Side effects associated with an increase in prolactin in the blood appear as dysmenorrhea or oligomenorrhea, pseudohermaphroditism in women, gynecomastia and delayed ejaculation in men, decreased libido, galactorrhea, hirsutism. In rare cases, there are changes in blood sugar levels, as well as symptoms of diabetes insipidus.
In rare cases, there are changes in blood sugar levels, as well as symptoms of diabetes insipidus.
Severe complications of neuroleptic therapy include general allergic and toxic reactions, hepatitis, pathological changes in the organ of vision (pathological pigmentation of the refractive media, combined with pathological pigmentation of the skin of the hands and face - "skin-eye syndrome", toxic changes in the retina), a violation of the blood picture (leukopenia , agranulocytosis, aplastic anemia, thrombocytopenia). Among the mental disorders associated with therapy, there are anesthetic depression, a painful disturbance in the sense of sleep, delirium (more often it occurs with a sharp change in doses of neuroleptics in people with organic diseases of the central nervous system, the elderly or children), epileptiform seizures.
Antipsychotics of new generations, compared with traditional derivatives of phenothiazines and butyrophenones, cause significantly fewer side effects and complications.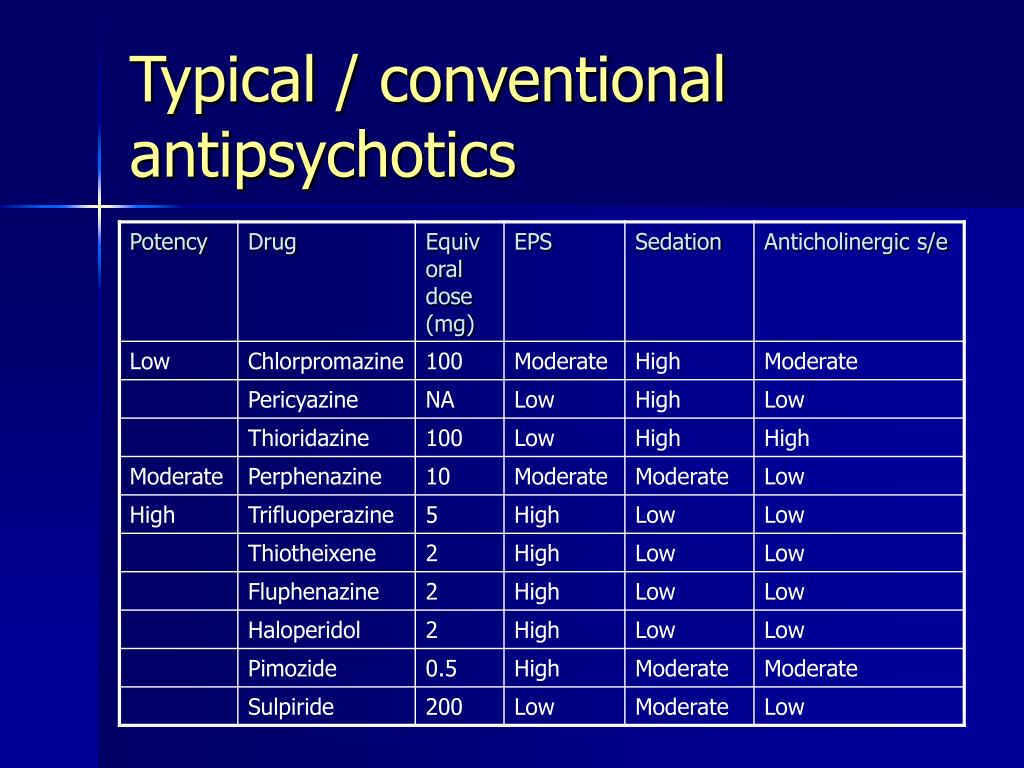
Antidepressants. Side effects related to the central nervous system and the autonomic nervous system are expressed as dizziness, tremor, dysarthria, impaired consciousness in the form of delirium, epileptiform seizures. Possible exacerbation of anxious disorders, activation of suicidal tendencies, inversion of affect, drowsiness or, conversely, insomnia. Side effects may be manifested by hypotension, sinus tachycardia, arrhythmia, impaired atrioventricular conduction. Complications from the hematopoietic system are relatively rare. Their clinical signs are depression of bone marrow function, leukopenia, agranulocytosis, thrombocytopenia, hemolytic anemia. Violation of the function of the endocrine system is limited to a change in the content of sugar in the blood (a downward trend).
In the treatment of antidepressants, there are also side effects such as dry mucous membranes, disturbance of accommodation, increased intraocular pressure, hypo- or atony of the intestine (constipation), urinary retention. More often they are observed when taking traditional tricyclic antidepressants and are associated with their anticholinergic effect. The use of tricyclic drugs is often accompanied by an increase in appetite and a significant increase in body weight. With the simultaneous use of MAO inhibitors with foods containing tyramine or its precursor - tyrosine (cheeses, etc.), a "cheese effect" occurs, manifested by hypertension, hyperthermia, convulsions and sometimes leading to death.
More often they are observed when taking traditional tricyclic antidepressants and are associated with their anticholinergic effect. The use of tricyclic drugs is often accompanied by an increase in appetite and a significant increase in body weight. With the simultaneous use of MAO inhibitors with foods containing tyramine or its precursor - tyrosine (cheeses, etc.), a "cheese effect" occurs, manifested by hypertension, hyperthermia, convulsions and sometimes leading to death.
Newer generations of antidepressants are better tolerated and safer. It can only be noted that when prescribing serotonin reuptake inhibitors and reversible MAO-A inhibitors, disturbances in the activity of the gastrointestinal tract (nausea, vomiting, diarrhea), headaches, insomnia, and anxiety are observed. The development of impotence while taking serotonin reuptake inhibitors has also been described. In cases of a combination of serotonin reuptake inhibitors with drugs of the tricyclic group, the formation of the so-called serotonin syndrome, manifested by fever and signs of intoxication. For tetracyclic antidepressants, daytime sleepiness and lethargy are more characteristic.
Tranquilizers. Side effects during treatment with tranquilizers are most often manifested by daytime drowsiness, lethargy,
muscle weakness, impaired concentration, short-term memory, as well as a slowdown in the speed of mental reactions. In some cases, paradoxical reactions develop in the form of anxiety, insomnia, psychomotor agitation, hallucinations. Ataxia, dysarthria, tremor occur much less frequently.
Among the dysfunctions of the autonomic nervous system and other organs and systems, hypotension, constipation, nausea, urinary retention or incontinence, decreased libido are noted. There may also be signs of depression of the respiratory center (respiratory arrest is possible). Changes in the functions of the organs of vision are manifested in the form of diplopia and disturbance of accommodation. Long-term use of tranquilizers is dangerous due to the possibility of developing addiction to them, i. e. mental and physical dependence.
Nootropics. Nootropic treatment side effects are rare. Sometimes there are nervousness, irritability, elements of psychomotor agitation and disinhibition of drives, as well as anxiety and insomnia. Possible dizziness, headache, tremor; in some cases, dyspeptic symptoms - nausea, abdominal pain.
Stimulants. These drugs have CNS side effects (tremor, euphoria, insomnia, irritability, headaches, and signs of psychomotor agitation). There may be disorders of the autonomic nervous system - sweating, dry mucous membranes, anorexia, as well as disorders of cardiovascular activity - arrhythmia, tachycardia, increased blood pressure. When treating patients with diabetes, it should be borne in mind that while taking stimulants, the body's sensitivity to insulin may change. It is also possible to have sexual dysfunction.
It is important to emphasize that prolonged and frequent use of stimulants can lead to the development of mental and physical dependence.
Lithium salts. Side effects or complications with the use of lithium salts usually occur at the beginning of therapy before a stable blood concentration of the drug is established. With proper therapy under the control of lithium in the blood and full informing the patient about the features of treatment, side effects rarely interfere with the prophylactic course. The patient must first of all be aware of the peculiarities of the diet - the exclusion of high intake of liquid and salt, the restriction of food rich in lithium - smoked meats, certain types of hard cheeses, red wine.
The most common side effect of lithium therapy is tremor. Severe tremor, indicative of the neurotoxic effect of lithium, increases with high plasma lithium concentrations. Often there are violations of the function of the gastrointestinal tract - nausea, vomiting, loss of appetite, diarrhea. Often there is an increase in body weight, polydipsia, polyuria. Lithium inhibits thyroid function, causing hypothyroidism. Usually these phenomena are transient. In severe cases, discontinuation of therapy is indicated. The consequences of lithium action on the cardiovascular system are similar to the picture of hypokalemia, but, as a rule, no special intervention is required. Acne, maculopapular rash, worsening of psoriasis are possible. There have been cases of alopecia. With prolonged lithium therapy, cognitive impairments may occur: memory loss, slowing down of psychomotor reactions, dysphoria. Signs of severe toxic conditions and overdose of drugs: metallic taste in the mouth, thirst, pronounced tremor, dysarthria, ataxia, and with a further increase in intoxication - impaired consciousness, fascicular muscle twitching, myoclonus, convulsions, coma. The longer the toxic level of lithium in the blood persists, the greater the likelihood of irreversible changes in the central nervous system, and in especially severe cases, death.
Usually these phenomena are transient. In severe cases, discontinuation of therapy is indicated. The consequences of lithium action on the cardiovascular system are similar to the picture of hypokalemia, but, as a rule, no special intervention is required. Acne, maculopapular rash, worsening of psoriasis are possible. There have been cases of alopecia. With prolonged lithium therapy, cognitive impairments may occur: memory loss, slowing down of psychomotor reactions, dysphoria. Signs of severe toxic conditions and overdose of drugs: metallic taste in the mouth, thirst, pronounced tremor, dysarthria, ataxia, and with a further increase in intoxication - impaired consciousness, fascicular muscle twitching, myoclonus, convulsions, coma. The longer the toxic level of lithium in the blood persists, the greater the likelihood of irreversible changes in the central nervous system, and in especially severe cases, death.
Lithium therapy is contraindicated in patients with impaired renal excretory function, with cardiovascular diseases (in the stage of decompensation), chronic diseases of the gastrointestinal tract (gastric and duodenal ulcers, etc. ), epilepsy, with conditions requiring a salt-free diet, during pregnancy, old age. A relative contraindication to the appointment of lithium preparations is a violation of the function of the thyroid gland.
), epilepsy, with conditions requiring a salt-free diet, during pregnancy, old age. A relative contraindication to the appointment of lithium preparations is a violation of the function of the thyroid gland.
Anticonvulsants. The most common side effects that occur during treatment with anticonvulsants, in particular carbamazepine, are functional disorders of the central nervous system - lethargy, drowsiness, dizziness, ataxia. Hyperreflexia, myoclonus, tremor can be observed much less frequently. The severity of these phenomena is significantly reduced with a smooth increase in doses. In the course of therapy, they usually disappear. Sometimes there are side effects such as nausea, vomiting, constipation or diarrhea, loss of appetite; possible development of hepatitis. Severe and rare (1 in 20,000) complications of carbamazepine therapy include white blood cell suppression. This drug should be used with caution in patients with cardiovascular pathology (it can help reduce intracardiac conduction), with glaucoma, prostate adenoma and diabetes. With an overdose of carbamazepine, drowsiness occurs, which can turn into stupor and coma; sometimes there are convulsions and dyskinesia of the facial muscles, functional disorders of the autonomic nervous system - hypothermia, depression of the respiratory and vasomotor centers (sinus tachycardia, arterial hypo- and hypertension). With a pronounced cardiotoxic effect of carbamazepine, an atrioventricular block may develop.
With an overdose of carbamazepine, drowsiness occurs, which can turn into stupor and coma; sometimes there are convulsions and dyskinesia of the facial muscles, functional disorders of the autonomic nervous system - hypothermia, depression of the respiratory and vasomotor centers (sinus tachycardia, arterial hypo- and hypertension). With a pronounced cardiotoxic effect of carbamazepine, an atrioventricular block may develop.
General principles of treatment for side effects and complications of psychopharmacotherapy
With pronounced side effects, the question inevitably arises about the ratio of positive and negative effects of a particular drug and the advisability of continuing therapy. If the effectiveness of the drug clearly exceeds its undesirable effect, then in some cases it is enough to temporarily reduce the dose and then slowly increase it to the therapeutic one to improve its tolerability. Sometimes it is advisable to change the mode and rhythm of taking the drug with the redistribution of the daily dose during the day.
Often, additional therapeutic agents are required to stop side effects.
Special therapy, as a rule, is carried out in the presence of side effects caused by the use of antipsychotics. For the correction of extrapyramidal disorders most characteristic of neuroleptic therapy, anticholinergic drugs are used - trihexyphenidyl (artan, cyclodol, parkopan), bentropine (cogentin, tremblex), biperidine (akineton). Correctors of different pharmacological groups have different features of action, therefore, if the effectiveness of drugs from one group is low, drugs from another group should be prescribed or the form of administration should be changed (oral to parenteral). It must be remembered that corrective therapy should be started after the first signs of side effects, but not prophylactically.
In some patients, the desired effectiveness of therapy is achieved by a combination of anticholinergics with tranquilizers with a pronounced muscle relaxant effect (diazepam, lorazepam). A combination of anticholinergics, tranquilizers and β-blockers (propranolol) is indicated in the treatment of akathisia.
A combination of anticholinergics, tranquilizers and β-blockers (propranolol) is indicated in the treatment of akathisia.
A special tactic of treatment is carried out with tardive dyskinesia. According to modern concepts, the biological basis of these conditions is an increase in sensitivity and an increase in the density of dopaminergic receptors in the striatum due to their long-term blockade by antipsychotics. Based on this, it is recommended to reduce the doses of neuroleptics. If the phenomena of tardive dyskinesia increase, then neuroleptics must be canceled. A positive effect in these cases is sometimes given by the appointment of GABA agonists (baclofen, aminalon, picamilon), agonists of cholinergic receptors (tacrine, cogitum), B vitamins. Some features are observed only when orthostatic hypotension is relieved. For this purpose, it is recommended to use α-adrenergic stimulants, such as mezaton.
The tremor that appears at high concentrations of lithium in the blood is stopped by reducing the daily dose of the drug.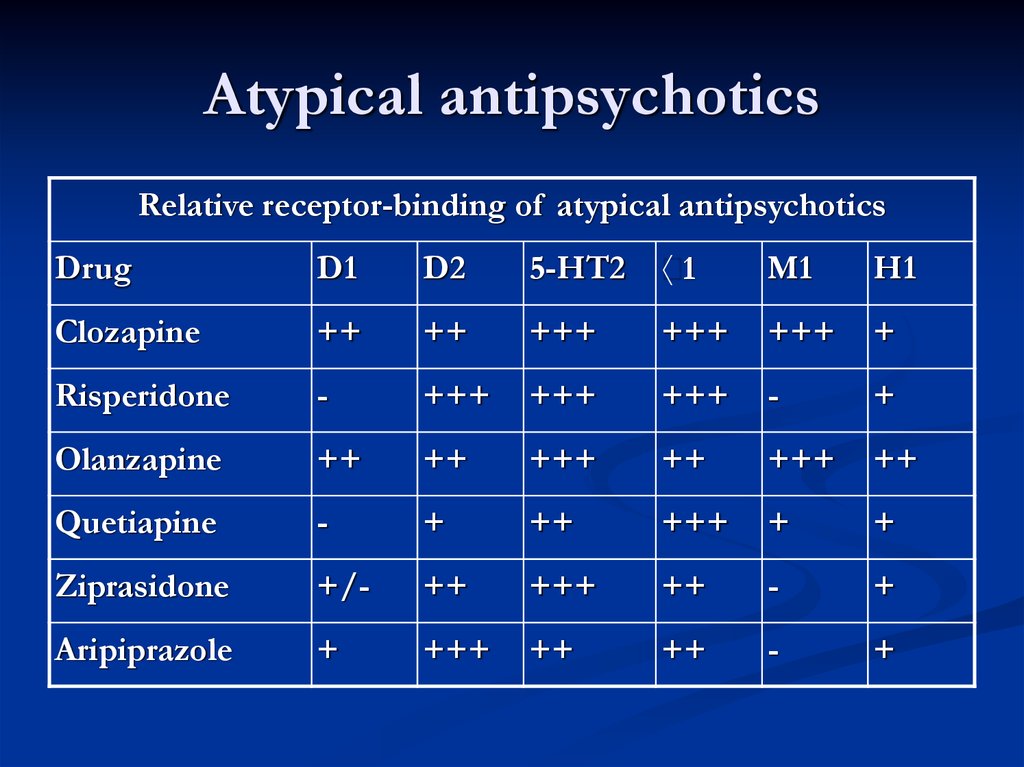 You can use a fractional division of the daily dose, as well as additionally prescribe antipsychotics. In the event of gastrointestinal disorders, lithium tolerance is improved by reducing doses, taking the drug in fractions, or taking it immediately after meals. These disorders, as well as weight gain, polydipsia, can also be corrected by the appointment of prolonged forms of lithium preparations, which allow you to smooth out sharp fluctuations in the concentration of the drug in the blood (the latter, according to existing ideas, cause the occurrence of side effects). In severe disorders of the thyroid gland, lithium therapy is stopped and hormonal preparations are prescribed. With severe intoxication, lithium preparations should be immediately canceled and dehydration should be combated; in severe cases, hemodialysis is indicated.
You can use a fractional division of the daily dose, as well as additionally prescribe antipsychotics. In the event of gastrointestinal disorders, lithium tolerance is improved by reducing doses, taking the drug in fractions, or taking it immediately after meals. These disorders, as well as weight gain, polydipsia, can also be corrected by the appointment of prolonged forms of lithium preparations, which allow you to smooth out sharp fluctuations in the concentration of the drug in the blood (the latter, according to existing ideas, cause the occurrence of side effects). In severe disorders of the thyroid gland, lithium therapy is stopped and hormonal preparations are prescribed. With severe intoxication, lithium preparations should be immediately canceled and dehydration should be combated; in severe cases, hemodialysis is indicated.
Complications in the treatment of carbamazepine can also be stopped by reducing the dose and prevent their development by smoothly increasing the dose. In more severe cases, with an overdose of carbamazepine, you should immediately wash the stomach, prescribe activated charcoal, and then carry out intensive general medical detoxification measures.
In more severe cases, with an overdose of carbamazepine, you should immediately wash the stomach, prescribe activated charcoal, and then carry out intensive general medical detoxification measures.
Thus, a large group of side effects of psychotropic drugs does not require special therapy, since their severity decreases with continued therapy with the same doses or with a decrease in doses. This applies, in particular, to the sedative effect and orthostatic disorders. With a significant severity of side effects and the inability to cancel therapy or transfer the patient to other drugs, appropriate symptomatic agents are prescribed and treatment is continued under the constant supervision of a doctor with laboratory parameters monitored.
Influence of side effects and complications caused by atypical antipsychotics on the effectiveness of therapy in patients with schizophrenia
It is well known that antipsychotic therapy is accompanied by the development of side effects and complications from various body systems. Numerous studies are devoted to their description, risk assessment, study of development mechanisms and development of correction methods. Some complications that develop acutely due to increased individual sensitivity (malignant neuroleptic syndrome, agranulocytosis, torsades de pointes, myocarditis, etc.) pose a threat to the health and life of patients [8, 9, 15, 18, 26]. Side effects play a role in the development of chronic somatic diseases [3, 4, 19, 20, 24]. Adverse events cause subjective discomfort in patients, leading to a violation of the therapy regimen [5-7, 23, 25]. These violations can significantly reduce the effectiveness of treatment. However, this issue has been little studied.
Numerous studies are devoted to their description, risk assessment, study of development mechanisms and development of correction methods. Some complications that develop acutely due to increased individual sensitivity (malignant neuroleptic syndrome, agranulocytosis, torsades de pointes, myocarditis, etc.) pose a threat to the health and life of patients [8, 9, 15, 18, 26]. Side effects play a role in the development of chronic somatic diseases [3, 4, 19, 20, 24]. Adverse events cause subjective discomfort in patients, leading to a violation of the therapy regimen [5-7, 23, 25]. These violations can significantly reduce the effectiveness of treatment. However, this issue has been little studied.
The aim of this study was to evaluate the significance of adverse events associated with the use of clozapine, risperidone, olanzapine and quetiapine for the effectiveness of the treatment of schizophrenia.
The objectives of the study were as follows: 1) to assess the frequency and severity of adverse events in the treatment of schizophrenia and compare the results with literature data; 2) to analyze the mechanisms of the relationship between tolerance and the effectiveness of the relief therapy for schizophrenia; 3) to determine the main types of adverse events that reduce the effectiveness of the relief therapy of schizophrenia; 4) to determine the features of the mental state of patients in which a decrease in the effectiveness of stopping therapy due to its poor tolerance is most likely; 5) to compare the importance of side effects and complications in limiting the possibility of effective treatment with various atypical antipsychotics; 6) based on the analysis of the data obtained, propose a classification of side effects and complications that reduce the effectiveness of the relief therapy for schizophrenia.
When studying the question posed in the study, we did not dwell in detail on determining the frequency and analysis of the dynamics of side effects of antipsychotic therapy, since these data are presented in detail in the literature [5, 11, 13, 16, 17, 22].
Material and methods
We examined 434 patients with schizophrenia occurring with a predominance of productive symptoms in a state of exacerbation or acute attacks of the disease.
Patients were divided into 5 groups depending on the atypical neuroleptic treated: clozapine (group 1), risperidone (group 2), olanzapine (group 3) or quetiapine (group 4), and in paranoid schizophrenia - also with highly potent typical antipsychotics (group 5). Their characteristics are presented in table. 1.
Cupping therapy was usually carried out in a hospital. In some cases (initial manifestations of acute psychoses, subacute conditions, “erased” exacerbations of continuous paranoid schizophrenia, exacerbations of a sluggish form), treatment could be carried out on an outpatient basis. Doses of neuroleptics were selected individually, taking into account the severity of the condition and its dynamics. The duration of follow-up for schizophrenia occurring in the form of seizures and sluggish - was 10 weeks (2.5 months), for paranoid - 12 weeks (3 months).
Doses of neuroleptics were selected individually, taking into account the severity of the condition and its dynamics. The duration of follow-up for schizophrenia occurring in the form of seizures and sluggish - was 10 weeks (2.5 months), for paranoid - 12 weeks (3 months).
Adverse events (side effects, complications) developing during therapy were assessed clinically and, if necessary, their severity was assessed using the UKU scale [12, 21]. Clinical and biochemical blood tests, urinalysis, ECG were studied. If necessary, other laboratory and instrumental examination methods were used. The influence of adverse events on the effectiveness of the treatment process was analyzed. The risk to the health (life) of patients was assessed, the number of patients in whom side effects and complications caused a change in the treatment regimen (by the doctor or the patient himself) with a loss of its effectiveness was taken into account.
Statistical analysis of the obtained data was carried out using the methods of parametric and non-parametric statistics (calculation of 95% confidence interval for average values, calculation of the value " p " using the methods "χ 2 ", "χ 2 " with Yates correction or two-sided Fisher's exact test).
Results
The frequency of the main adverse events in the treatment of clozapine, risperidone, olanzapine, quetiapine and typical neuroleptics, identified in this study, is presented in table. 2. It corresponded to the results obtained earlier. None of the patients, when assessed on the UKU scale, had side effects of "severe degree" (3 points) lasting more than 10 days. This was due to the timely correction of therapy (change in the dose of neuroleptic, appointment of correctors).
The focus of this study was on studying the impact of developing adverse events during therapy on its effectiveness. As the analysis of the studied material showed, side effects and complications could significantly reduce the effectiveness of the treatment process due to the need to revise the treatment regimen. Firstly, some adverse events posed a certain kind of threat to the somatic condition of patients. In these cases, it was necessary to revise the treatment regimen according to "vital indications".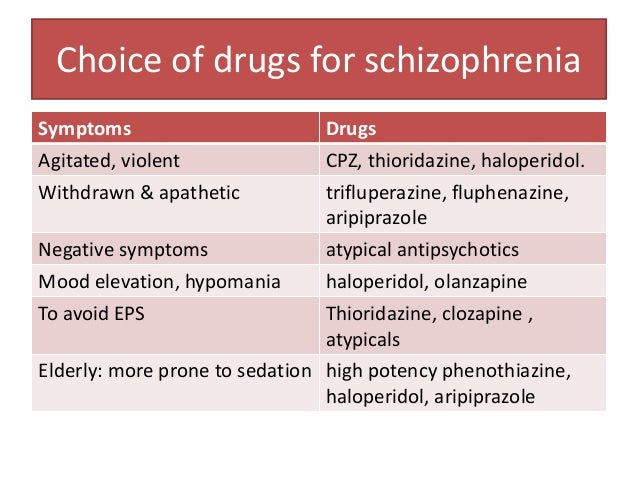 Secondly, with the development of side effects, patients could experience discomfort to one degree or another, worsening their general condition. These adverse events did not pose a health hazard, but their occurrence sometimes required a revision of the treatment regimen for ethical reasons. The effectiveness of the treatment process in this case also decreased due to the difficulty in the formation of therapeutic cooperation, the negative attitude of patients to taking medications. Thirdly, side effects could cause a negative attitude towards the treatment of relatives, their interference in the therapy process, and even refusal to continue it.
Secondly, with the development of side effects, patients could experience discomfort to one degree or another, worsening their general condition. These adverse events did not pose a health hazard, but their occurrence sometimes required a revision of the treatment regimen for ethical reasons. The effectiveness of the treatment process in this case also decreased due to the difficulty in the formation of therapeutic cooperation, the negative attitude of patients to taking medications. Thirdly, side effects could cause a negative attitude towards the treatment of relatives, their interference in the therapy process, and even refusal to continue it.
The complications noted in the study that are dangerous to health (life) developed as a consequence of typical side effects of neuroleptics, which in themselves did not pose a threat. Such complications in the surveyed sample included pneumonia and orthostatic collapse. Other complications, incl. developing due to increased individual sensitivity (neuroleptic malignant syndrome, damage to the blood, liver, heart, etc. ), was not observed, possibly due to the relatively small number of patients examined.
), was not observed, possibly due to the relatively small number of patients examined.
The development of pneumonia was noted in 4 patients taking clozapine (3.8%). They were all prescribed high doses of antipsychotics (400-550 mg/day). These patients belonged to the group of schizophrenia occurring in the form of seizures, and their condition was characterized by advanced acute psychoses with pronounced agitation, negativism, aggression, and refusal to accept medical care. In all cases, a rapid increase in the dose of neuroleptic was carried out within 2-5 days to the indicated above. Pneumonia manifested itself on the 5th - 7th days of therapy. It developed according to aspiration and hypostatic mechanisms due to aspiration of saliva with severe hypersalivation (2-3 points on the UKU scale) and limitation of patients' mobility due to prolonged drug-induced sleep or mechanical fixation. Carrying out specific treatment in all cases led to a rapid resolution of the complication without any further consequences, incl. remote, for the health of patients. When taking risperidone, olanzapine, quetiapine or typical antipsychotics, the development of pneumonia was not observed. There were no statistically significant differences between the groups, which, however, could be due to the rarity of this complication with the relative small number of the studied sample. It should be noted that previously only a few authors reported the possibility of developing pneumonia during therapy with typical antipsychotics [1]. The mechanism of its development was associated with a decrease in the motor activity of patients, impaired swallowing and cough reflex, but not with hypersalivation. Observations of pneumonia when taking atypical antipsychotics are not reported in the literature.
remote, for the health of patients. When taking risperidone, olanzapine, quetiapine or typical antipsychotics, the development of pneumonia was not observed. There were no statistically significant differences between the groups, which, however, could be due to the rarity of this complication with the relative small number of the studied sample. It should be noted that previously only a few authors reported the possibility of developing pneumonia during therapy with typical antipsychotics [1]. The mechanism of its development was associated with a decrease in the motor activity of patients, impaired swallowing and cough reflex, but not with hypersalivation. Observations of pneumonia when taking atypical antipsychotics are not reported in the literature.
Orthostatic collapse was observed in 7 (6.7%) patients taking clozapine, 1 (0.96%) risperidone, 2 (2.1%) olanzapine, 2 (2.1%) quetiapine, and in 1 (3.2%) case of therapy with typical antipsychotics (zuclopenthixol). No statistically significant differences were found between the groups. In all patients, syncope was observed once. Loss of consciousness lasted for 30-90 s. An indicator of the short duration of these conditions was the fact that the time of loss of consciousness was insufficient to take measures to measure blood pressure. Orthostatic collapses developed on days 2-5 of therapy against the background of a rapid increase in the dose of antipsychotics. They were preceded by complaints of dizziness, weakness, flies, lightheadedness or nausea that appeared when the body position changed (2-3 points on the UKU scale). Orthostatic syncope was observed both in acute psychoses and exacerbations of indolent or paranoid schizophrenia (but in all cases with a short duration of the disease and a history of its treatment).
In all patients, syncope was observed once. Loss of consciousness lasted for 30-90 s. An indicator of the short duration of these conditions was the fact that the time of loss of consciousness was insufficient to take measures to measure blood pressure. Orthostatic collapses developed on days 2-5 of therapy against the background of a rapid increase in the dose of antipsychotics. They were preceded by complaints of dizziness, weakness, flies, lightheadedness or nausea that appeared when the body position changed (2-3 points on the UKU scale). Orthostatic syncope was observed both in acute psychoses and exacerbations of indolent or paranoid schizophrenia (but in all cases with a short duration of the disease and a history of its treatment).
With the development of pneumonia and orthostatic collapse, antipsychotic therapy was not canceled, and there was no “change” of antipsychotic. Only a temporary (from several days to 2 weeks) reduction of its daily dose was carried out according to "vital indications". This was enough to reduce the severity of hypersalivation, sedative and hypotensive effects. Subsequently, the doses of neuroleptics increased again, but more slowly. The re-described complications did not develop. It is important that the development of the described complications was to a large extent due to insufficient attention to the condition of sick doctors and medical personnel, since their development could be predicted in advance based on patients' complaints or with objective observation. This was confirmed by the absence of cases of persistent intestinal paresis or intestinal obstruction - complications resulting from the influence of antipsychotics with anticholinergic activity on the gastrointestinal tract. This is due to the fact that the medical staff and doctors were more attentive to this problem and took the necessary measures in a timely manner.
This was enough to reduce the severity of hypersalivation, sedative and hypotensive effects. Subsequently, the doses of neuroleptics increased again, but more slowly. The re-described complications did not develop. It is important that the development of the described complications was to a large extent due to insufficient attention to the condition of sick doctors and medical personnel, since their development could be predicted in advance based on patients' complaints or with objective observation. This was confirmed by the absence of cases of persistent intestinal paresis or intestinal obstruction - complications resulting from the influence of antipsychotics with anticholinergic activity on the gastrointestinal tract. This is due to the fact that the medical staff and doctors were more attentive to this problem and took the necessary measures in a timely manner.
It must be reiterated that all cases of the listed complications ended favorably. The use of specific methods of treatment led to a rapid improvement in the condition of patients.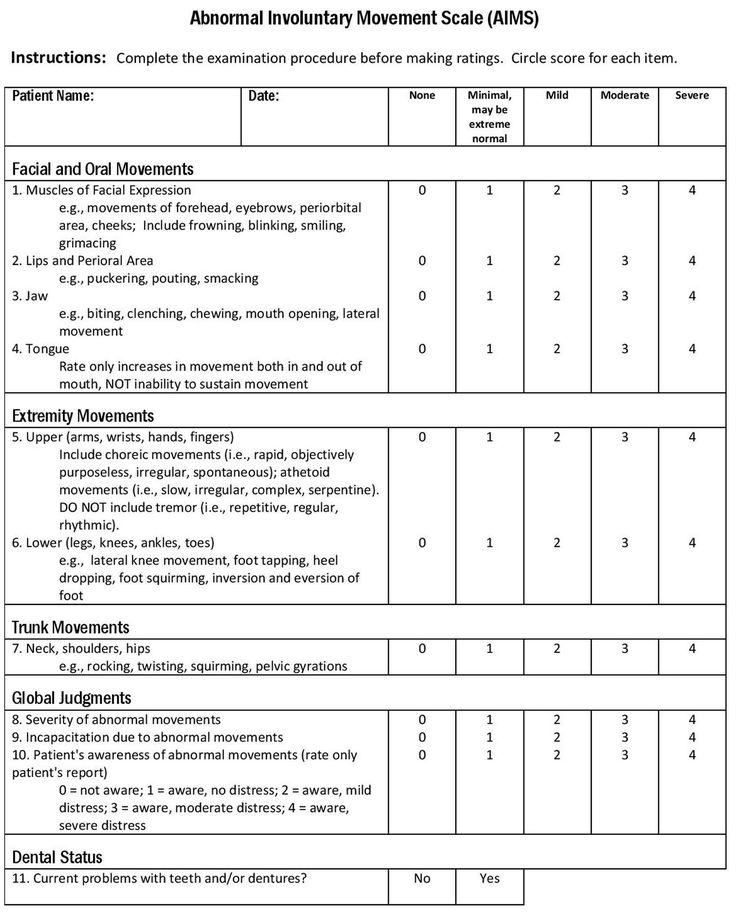 No long-term effects were noted. Perhaps this is due to the fact that most of the patients were young and somatically healthy. It can be assumed that with concomitant somatic pathology or in elderly patients, the described adverse events would not be so painless, as previously indicated by a number of authors [10, 14].
No long-term effects were noted. Perhaps this is due to the fact that most of the patients were young and somatically healthy. It can be assumed that with concomitant somatic pathology or in elderly patients, the described adverse events would not be so painless, as previously indicated by a number of authors [10, 14].
Another factor (in addition to complications) limiting the effectiveness of relief therapy was the appearance in patients of subjective discomfort caused by poor tolerability of antipsychotics. It was determined by the typical side effects presented in Table. 1. During treatment with clozapine, these effects included increased salivation, constipation and dizziness. The greatest discomfort was caused by severe drowsiness observed in the first weeks of therapy. In patients with agitation or anxiety, it certainly acted as a therapeutic effect, but the patients themselves were usually considered as a painful undesirable phenomenon. Negative perception of drowsiness was characteristic of all patients, but its consequences were different. With a slight severity of productive disorders (usually in patients with preserved criticism or at least consciousness of the disease), rational psychotherapy by methods of persuasion and indirect suggestion usually made it possible to maintain therapeutic cooperation in full. In cases of severe productive symptomatology and lack of criticism, therapeutic cooperation was usually broken. Patients tried to actively oppose medical care, although this was not so significant in a hospital setting.
With a slight severity of productive disorders (usually in patients with preserved criticism or at least consciousness of the disease), rational psychotherapy by methods of persuasion and indirect suggestion usually made it possible to maintain therapeutic cooperation in full. In cases of severe productive symptomatology and lack of criticism, therapeutic cooperation was usually broken. Patients tried to actively oppose medical care, although this was not so significant in a hospital setting.
Uncomfortable side effects of risperidone include nasal congestion and dizziness (usually in elderly patients or young women with sluggish schizophrenia). However, most often discomfort or fear was caused by involuntary movements. It is necessary to pay attention to the rather frequent development when taking risperidone (usually at a dose of 4 mg / day and above) of extrapyramidal symptoms in the form of acute dystonia or akathisia, which significantly exceeds its risk when treated with other atypical neuroleptics (see Table 1).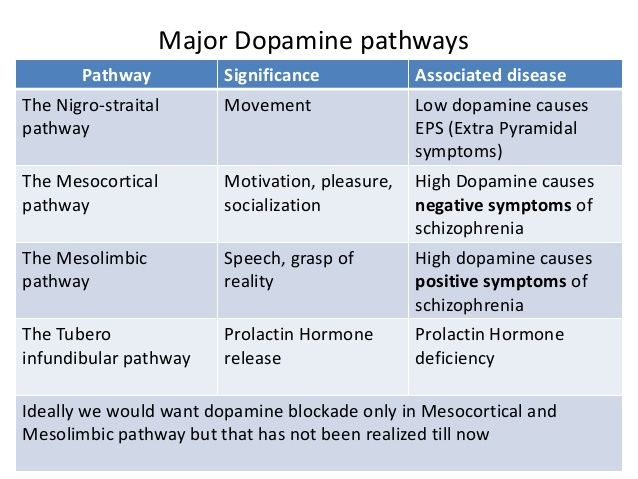
Patients treated with olanzapine most commonly complained of drowsiness, dizziness, dry mouth and constipation. Therapy with quetiapine led to the development of drowsiness and dizziness. Taking traditional antipsychotics most often caused the appearance of uncontrolled movements, constipation, and sometimes dizziness.
Most cases of adverse events that cause discomfort (with the exception of hypersalivation and orthostatic hypotension) were not dangerous. Perhaps this was due to the predominance of young somatically healthy patients in the examined group, careful monitoring of their condition to prevent the development of complications, and rational selection of doses of neuroleptics. However, severe discomfort sometimes required a revision of the treatment regimen for ethical reasons with a loss of its effectiveness. In the study, the change in the treatment regimen was limited to a decrease in the doses of antipsychotics. There were no cases of "change" of antipsychotics.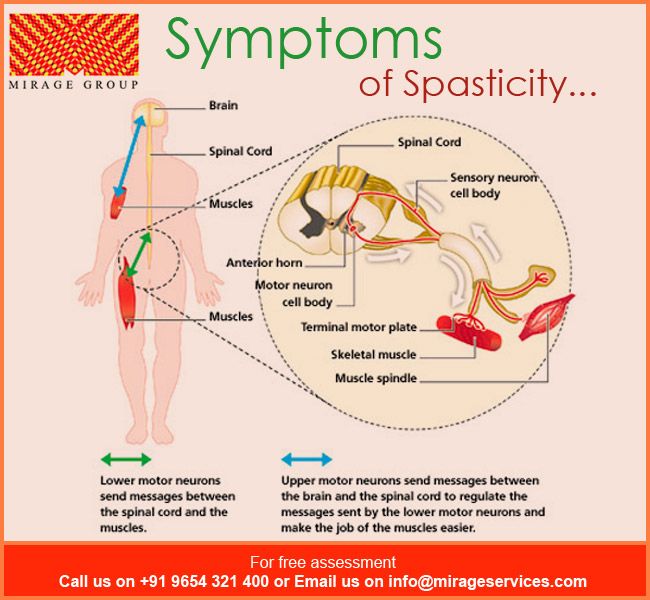 The number of observations of dose reduction of antipsychotics was small and amounted to 6 (5.7%), 3 (2.9%), 5 (5.2%), 0 and 3 (9.7%) patients treated with clozapine, risperidone, olanzapine, quetiapine, and typical agents (except in cases of dose reduction for pneumonia and collapse). Despite the fact that in the treatment of clozapine and typical antipsychotics, the number of such patients was somewhat greater than when taking other drugs, this difference reached statistical significance only in comparison with the group of patients taking quetiapine ( p = 0.03, Fisher's method and p = 0.01, Fisher method).
The number of observations of dose reduction of antipsychotics was small and amounted to 6 (5.7%), 3 (2.9%), 5 (5.2%), 0 and 3 (9.7%) patients treated with clozapine, risperidone, olanzapine, quetiapine, and typical agents (except in cases of dose reduction for pneumonia and collapse). Despite the fact that in the treatment of clozapine and typical antipsychotics, the number of such patients was somewhat greater than when taking other drugs, this difference reached statistical significance only in comparison with the group of patients taking quetiapine ( p = 0.03, Fisher's method and p = 0.01, Fisher method).
Reducing the doses of antipsychotics in all cases was carried out during the first 2-4 weeks of therapy, since in the future the severity of most subjectively unpleasant adverse reactions weakened spontaneously. Such “spontaneous” attenuation of the severity of neurological and somatovegetative adverse events (development of adaptation) is well known and has been described in the literature previously [1, 2, 5, 13].
Subjective discomfort caused by therapy could influence the attitude of patients towards it. This was manifested by a decrease in patients' confidence in the doctor, the appearance or strengthening of their negative attitude towards drugs, and attempts to violate the therapy regimen. The latter factor, however, did not play an important role in inpatient treatment, but was of great importance if the therapy was carried out on an outpatient basis. In some patients, the discomfort caused by relief therapy was reflected in therapeutic cooperation during the period of subsequent maintenance treatment. This category included patients with a low quality of formed remissions, especially with their paranoid variant as part of various forms of the disease, with a predominance of neurotic hypochondria or psychopathic symptoms in the structure of the state. In patients with a high quality of remissions, subjective discomfort was usually less reflected in therapeutic cooperation with further maintenance treatment. In this regard, the most indicative cases are acute (or rather "acute") psychoses, in which the discomfort from taking antipsychotics at the beginning of relief therapy sharply increased negativism, protest against treatment, violated the patients' trust in the doctor, up to a complete refusal to communicate. However, with the weakening of psychopathological symptoms, the emergence of consciousness of the disease, and then criticism, the formation of a full-fledged therapeutic cooperation was observed with a high level of trust and correct adherence to the medication regimen, despite the persisting discomfort.
In this regard, the most indicative cases are acute (or rather "acute") psychoses, in which the discomfort from taking antipsychotics at the beginning of relief therapy sharply increased negativism, protest against treatment, violated the patients' trust in the doctor, up to a complete refusal to communicate. However, with the weakening of psychopathological symptoms, the emergence of consciousness of the disease, and then criticism, the formation of a full-fledged therapeutic cooperation was observed with a high level of trust and correct adherence to the medication regimen, despite the persisting discomfort.
Cases of severe subjective discomfort due to individual intolerance to one of the antipsychotics or psychopharmacotherapy in general deserve special attention. In the first variant, the appointment of even small doses of neuroleptics led to the development of pronounced side effects that caused severe discomfort to patients. In our observation, there were 5 such patients (1. 2% of their total number). In 2 patients taking risperidone, severe forms of acute dystonia (3 points on the UKU scale) were observed in the form of oculogeric crises and pronounced torsion spasms. Risperidone doses were 2 and 4 mg/day. The development of dystonia required a reduction in the dosage of the antipsychotic for ethical reasons, since high doses of trihexyphenidyl (up to 16 mg/day) were ineffective. The mental state of these patients was characterized by "erased" exacerbations that developed against the background of a long course of fur-like schizophrenia with signs of transition to a continuous type. Two patients treated with olanzapine (7.5 mg/day and 10 mg/day) developed severe akathisia (UKU score 3) with a predominance of the anxiety component. These cases were referred to as sluggish schizophrenia. In 1 patient with paranoid schizophrenia who took quetiapine, two hard, painful formations the size of a penny in the area of one of the nipples were observed, but did not merge with it.
2% of their total number). In 2 patients taking risperidone, severe forms of acute dystonia (3 points on the UKU scale) were observed in the form of oculogeric crises and pronounced torsion spasms. Risperidone doses were 2 and 4 mg/day. The development of dystonia required a reduction in the dosage of the antipsychotic for ethical reasons, since high doses of trihexyphenidyl (up to 16 mg/day) were ineffective. The mental state of these patients was characterized by "erased" exacerbations that developed against the background of a long course of fur-like schizophrenia with signs of transition to a continuous type. Two patients treated with olanzapine (7.5 mg/day and 10 mg/day) developed severe akathisia (UKU score 3) with a predominance of the anxiety component. These cases were referred to as sluggish schizophrenia. In 1 patient with paranoid schizophrenia who took quetiapine, two hard, painful formations the size of a penny in the area of one of the nipples were observed, but did not merge with it. In this case, quetiapine therapy was canceled, however, not only because of this undesirable phenomenon, but also because of its low effectiveness. With further dynamic observation, the described formations disappeared within 2 weeks after the abolition of the antipsychotic. Cases of orthostatic collapses, which can be regarded as increased individual sensitivity of patients to the hypotensive effect, were discussed above. Also of interest are the observations of 4 patients in whom, according to the anamnesis, “the use of traditional antipsychotics was accompanied by the development of severe extrapyramidal symptoms with insufficient effectiveness of the use of correctors. When included in this study, they were prescribed risperidone (1 observation), olanzapine (1) and quetiapine (2). After the start of therapy, patients described the appearance of a subjectively painful sensation of “riveting” their eyes to one point or involuntary prolonged “examination of objects” within a limited area.
In this case, quetiapine therapy was canceled, however, not only because of this undesirable phenomenon, but also because of its low effectiveness. With further dynamic observation, the described formations disappeared within 2 weeks after the abolition of the antipsychotic. Cases of orthostatic collapses, which can be regarded as increased individual sensitivity of patients to the hypotensive effect, were discussed above. Also of interest are the observations of 4 patients in whom, according to the anamnesis, “the use of traditional antipsychotics was accompanied by the development of severe extrapyramidal symptoms with insufficient effectiveness of the use of correctors. When included in this study, they were prescribed risperidone (1 observation), olanzapine (1) and quetiapine (2). After the start of therapy, patients described the appearance of a subjectively painful sensation of “riveting” their eyes to one point or involuntary prolonged “examination of objects” within a limited area. After the appointment of correctors, there was a significant relief, which indicated the connection of the described symptom with extrapyramidal disorders.
After the appointment of correctors, there was a significant relief, which indicated the connection of the described symptom with extrapyramidal disorders.
A more severe variant of individual intolerance included cases of the development of pronounced side effects when prescribing various antipsychotics, i.e. psychopharmacotherapy in general. In such patients, its implementation was significantly difficult, since they experience severe discomfort when taking many antipsychotics. There were no such observations in the present study. However, individual intolerance to psychopharmacotherapy was established according to the anamnesis at the stage of inclusion in the study in 5 patients (1.2% of their total number). These observations were related to sluggish (4 observations) and paranoid (1) schizophrenia. In the past, patients have taken risperidone, olanzapine, flupentixol, zuclopenthixol, perphenazine, trifluoperazine, haloperidol, clozapine. All antipsychotics, even in small doses, caused the development of severe side effects that are difficult to correct.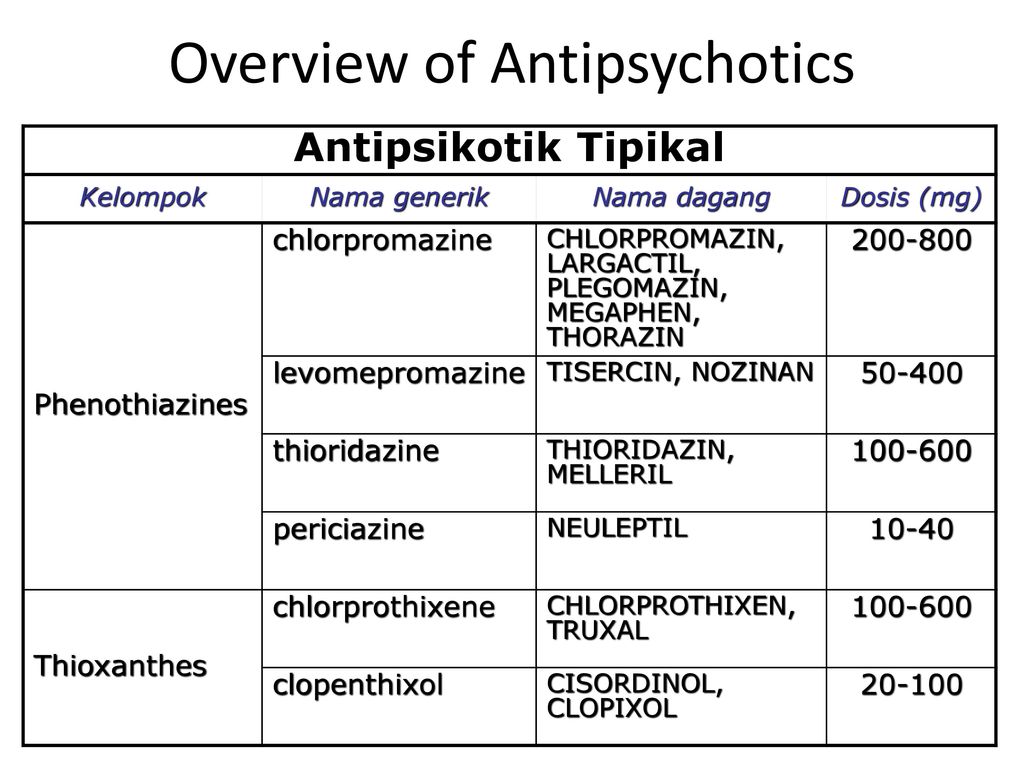 This led to the formation of a negative attitude of patients to taking medications and its break in the outpatient setting. In the present study, all patients were prescribed quetiapine based on literature data on its relatively favorable side effect profile. The therapy was well tolerated. With sluggish schizophrenia, she was successful. In a patient with a paranoid form, the effectiveness of treatment was low, but it was continued based on the consideration that the low effectiveness of treatment is more useful than its complete ineffectiveness when the patient refuses to take medication (benefit / risk ratio).
This led to the formation of a negative attitude of patients to taking medications and its break in the outpatient setting. In the present study, all patients were prescribed quetiapine based on literature data on its relatively favorable side effect profile. The therapy was well tolerated. With sluggish schizophrenia, she was successful. In a patient with a paranoid form, the effectiveness of treatment was low, but it was continued based on the consideration that the low effectiveness of treatment is more useful than its complete ineffectiveness when the patient refuses to take medication (benefit / risk ratio).
In a separate group, it is advisable to allocate patients in whom individual "intolerance" was manifested by a deterioration in mental state due to an unusual interpretation of side effects. In this case, it is necessary to note some conventionality of the term "intolerance", since it arose as a result of individual "psychopathological processing" of subjective discomfort caused by side effects.![]() There were 10 such patients (2.3%). 2 patients suffering from paranoid schizophrenia with its long course closer to non-remission, taking clozapine, perceived the therapy as "poisoning the body". These statements in the literal sense did not testify to the delirium of poisoning, but rather to delusional fantasies. In this way, they expressed their attitude to the ongoing therapy as "heavy", uncomfortable. Another 4 patients, 2 of whom took clozapine, and 2 - olanzapine, built "neuroleptic discomfort" into the delusional system and hid under the mask of sensory mental automatism and delusions of influence. Finally, in 4 patients (1 - risperidone, 1 - olanzapine and 2 - haloperidol) with delusional hypochondria, the unpleasant bodily sensations caused by taking antipsychotics were built into the system of hypochondriacal delirium. In all these cases, the increased severity of side effects led to a deterioration in the mental state of patients, despite effective therapy. The appearance of anxiety or arousal drew attention.
There were 10 such patients (2.3%). 2 patients suffering from paranoid schizophrenia with its long course closer to non-remission, taking clozapine, perceived the therapy as "poisoning the body". These statements in the literal sense did not testify to the delirium of poisoning, but rather to delusional fantasies. In this way, they expressed their attitude to the ongoing therapy as "heavy", uncomfortable. Another 4 patients, 2 of whom took clozapine, and 2 - olanzapine, built "neuroleptic discomfort" into the delusional system and hid under the mask of sensory mental automatism and delusions of influence. Finally, in 4 patients (1 - risperidone, 1 - olanzapine and 2 - haloperidol) with delusional hypochondria, the unpleasant bodily sensations caused by taking antipsychotics were built into the system of hypochondriacal delirium. In all these cases, the increased severity of side effects led to a deterioration in the mental state of patients, despite effective therapy. The appearance of anxiety or arousal drew attention. Moreover, in this case, it was impossible to talk about the imitation of psychopathological symptoms by side effects, as happens in the case of increased catatonic hyperkinesis in lucid catatonia or inaccessibility in paranoid schizophrenia when using high and ultra-high doses of antipsychotics. The described deterioration of the mental state was associated with "internal", "psychopathological" processing of subjectively painful bodily discomfort.
Moreover, in this case, it was impossible to talk about the imitation of psychopathological symptoms by side effects, as happens in the case of increased catatonic hyperkinesis in lucid catatonia or inaccessibility in paranoid schizophrenia when using high and ultra-high doses of antipsychotics. The described deterioration of the mental state was associated with "internal", "psychopathological" processing of subjectively painful bodily discomfort.
The possibility of conducting effective psychopharmacotherapy in a number of cases was limited by the fact that due to the development of side effects in patients, the negative attitude of their relatives to treatment arose or increased . There were 19 observations in which side effects played a large role in the formation of a negative attitude of relatives to treatment (4.4% of the number of patients examined). The side effects that most often caused a negative attitude of relatives included involuntary movements, severe drowsiness and lethargy, salivation. However, since relatives of a certain psychological type protested against the treatment, such a reaction could have been caused even by patients' complaints of minor discomfort. Usually, with the initial pretentiousness of their statements, a conversation, a discussion of the meaning and consequences of adverse events, calmed family members. However, sometimes, despite these measures, relatives actively intervened in the therapy process or refused to carry it out. In these cases, it was necessary to actively use the methods of psychoeducation and rational psychotherapy of relatives, incl. with the involvement of other doctors, which made it possible to continue the relief therapy in all cases. However, during the period of further maintenance therapy, when the doctor's control over its implementation was significantly weakened, the negative attitude of relatives towards it led to a rapid interruption of treatment.
However, since relatives of a certain psychological type protested against the treatment, such a reaction could have been caused even by patients' complaints of minor discomfort. Usually, with the initial pretentiousness of their statements, a conversation, a discussion of the meaning and consequences of adverse events, calmed family members. However, sometimes, despite these measures, relatives actively intervened in the therapy process or refused to carry it out. In these cases, it was necessary to actively use the methods of psychoeducation and rational psychotherapy of relatives, incl. with the involvement of other doctors, which made it possible to continue the relief therapy in all cases. However, during the period of further maintenance therapy, when the doctor's control over its implementation was significantly weakened, the negative attitude of relatives towards it led to a rapid interruption of treatment.
In tab. 3. summarized data on the number of patients who, for the reasons listed above, had a reduction in the dose of antipsychotics are presented. This was most frequently required with clozapine and conventional agents, and least frequently with quetiapine. Noteworthy is the absence of statistically significant differences between groups of patients treated with olanzapine, clozapine, and typical antipsychotics. A significant difference was not determined when comparing groups of patients taking olanzapine, risperidone and quetiapine. These observations allow us to conclude that the importance of side effects in limiting the possibility of effective treatment decreases in the series of typical antipsychotics and clozapine-olanzapine-risperidone and quetiapine.
This was most frequently required with clozapine and conventional agents, and least frequently with quetiapine. Noteworthy is the absence of statistically significant differences between groups of patients treated with olanzapine, clozapine, and typical antipsychotics. A significant difference was not determined when comparing groups of patients taking olanzapine, risperidone and quetiapine. These observations allow us to conclude that the importance of side effects in limiting the possibility of effective treatment decreases in the series of typical antipsychotics and clozapine-olanzapine-risperidone and quetiapine.
Talk
It can be seen from the above that adverse events that develop with the use of atypical and traditional antipsychotics reduce the effectiveness of relief therapy, limiting the possibility of its implementation. In these cases, it becomes necessary to revise the treatment regimen for "vital indications" (due to the development of dangerous complications), ethical considerations to maintain therapeutic cooperation (in case of subjective discomfort experienced by patients) or at the insistence of relatives (due to their negative attitude towards therapy, usually with side effects).
Complications dangerous to health (life) during antipsychotic therapy are quite rare. In the studied sample, there were cases of the development of pneumonia when taking clozapine and orthostatic collapses in the treatment of all antipsychotics. These adverse reactions develop as a consequence of typical side effects (hypersalivation, sedation, orthostatic hypotension). Insufficiently careful attention to the condition of patients is of great importance in their development, since it is possible to predict the risk of their development in advance by complaints and with objective observation. For young somatically healthy patients, the listed complications do not pose a threat, provided that they are adequately given specific therapy, careful dynamic monitoring and rehabilitation measures.
Many typical side effects cause subjective discomfort in patients. Its role in limiting the possibility of effective treatment is determined by ethical factors (easing the suffering of the patient) or practical considerations (maintaining therapeutic cooperation). In the latter case, when considering the issue of revising the treatment regimen, it is necessary to focus not only on the tolerability of therapy, but also on the severity of the patient's mental state. Patients with severe productive symptoms and lack of criticism perceive side effects more negatively than patients with preserved criticism or consciousness of the disease. With cupping therapy in a hospital, the possibility of a violation of the medication regimen is small, but it must be borne in mind that the patient will take medications on an outpatient basis in the future. If there is a prospect of a significant improvement in the mental state, the subjectively painful side effects that develop during stopping therapy can be neglected to a certain extent. On the contrary, if the improvement is expected to be insignificant, the treatment regimen should be reviewed to avoid the patient's refusal of psychiatric care, incl. receiving neuroleptics, outside the hospital. In these cases, it is better to prescribe another neuroleptic, albeit less effective, or to reduce its dose, guided by the principle: the low effectiveness of treatment is more useful than the complete lack of effectiveness if the patient refuses it.
In the latter case, when considering the issue of revising the treatment regimen, it is necessary to focus not only on the tolerability of therapy, but also on the severity of the patient's mental state. Patients with severe productive symptoms and lack of criticism perceive side effects more negatively than patients with preserved criticism or consciousness of the disease. With cupping therapy in a hospital, the possibility of a violation of the medication regimen is small, but it must be borne in mind that the patient will take medications on an outpatient basis in the future. If there is a prospect of a significant improvement in the mental state, the subjectively painful side effects that develop during stopping therapy can be neglected to a certain extent. On the contrary, if the improvement is expected to be insignificant, the treatment regimen should be reviewed to avoid the patient's refusal of psychiatric care, incl. receiving neuroleptics, outside the hospital. In these cases, it is better to prescribe another neuroleptic, albeit less effective, or to reduce its dose, guided by the principle: the low effectiveness of treatment is more useful than the complete lack of effectiveness if the patient refuses it.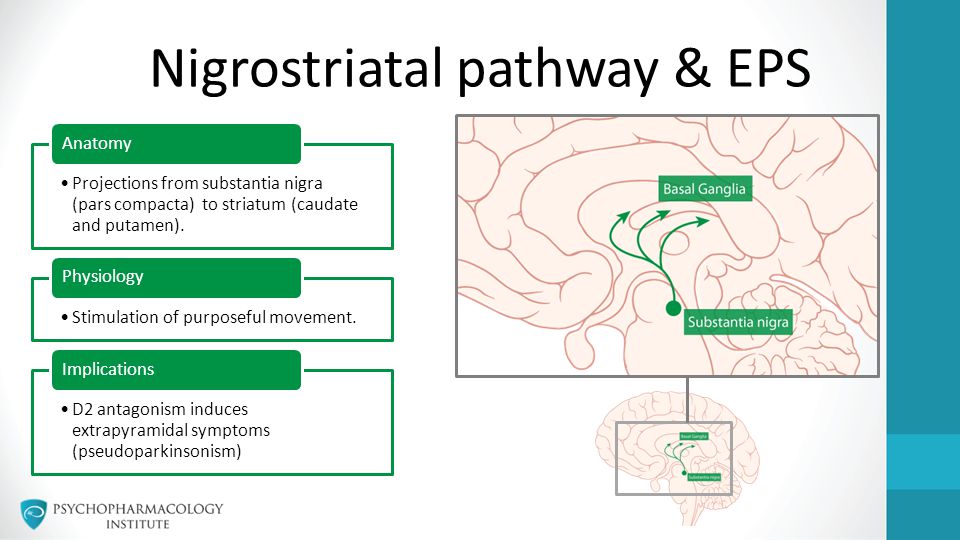 It must be taken into account that the tolerability of therapy improves significantly within a few weeks after the start of its administration due to the phenomenon of adaptation to side effects. Therefore, subjective discomfort weakens over time.
It must be taken into account that the tolerability of therapy improves significantly within a few weeks after the start of its administration due to the phenomenon of adaptation to side effects. Therefore, subjective discomfort weakens over time.
The most therapeutically "difficult" group consists of patients with individual intolerance to antipsychotics and patients whose side effects worsen their mental state due to their unusual interpretation, "psychopathological processing". In these cases, the effectiveness of the treatment process is significantly reduced due to the impossibility of prescribing adequate effective therapy, both for ethical reasons (to a large extent!), And from the standpoint of maintaining therapeutic cooperation. After "enumeration" of various antipsychotics, it is advisable to continue therapy with the drug with the best tolerance, even at the expense of the effectiveness of treatment.
The development of side effects may cause resistance to medical intervention by relatives of patients. In most cases, a simple conversation helps to avoid this. However, with a certain category of relatives, in order to maintain therapeutic cooperation, it is necessary to actively conduct psychotherapy and psychoeducation.
In most cases, a simple conversation helps to avoid this. However, with a certain category of relatives, in order to maintain therapeutic cooperation, it is necessary to actively conduct psychotherapy and psychoeducation.
The significance of side effects and complications in limiting the possibility of effective treatment decreases in the series of typical antipsychotics and clozapine-olanzapine-risperidone and quetiapine.
Based on the data obtained, it is possible to propose a classification of adverse events (side effects and complications) that reduce the effectiveness of the relief therapy for schizophrenia, or rather limit the possibility of its implementation (Table 4).
In conclusion, it should be noted that the present study was conducted in a university clinic with the possibility of a thorough individual approach to their treatment (careful follow-up, timely correction of therapy, psychotherapy of patients and their relatives). It can be assumed that under the conditions of normal clinical practice, the impact of side effects on the effectiveness of the treatment process may be more serious.