Do lamictal side effects go away
Side effects of lamotrigine - NHS
Like all medicines, lamotrigine can cause side effects, although not everyone gets them.
Most side effects of lamotrigine wear off, but it can take several weeks or months.
When you start taking lamotrigine, it's important to increase the dose slowly as this will help reduce or stop some side effects happening.
Lamotrigine can also cause some unpredictable side effects no matter what dose you take, and these can happen at any time.
Speak to your pharmacist or doctor if you're concerned about side effects.
Common side effects
These common side effects of lamotrigine may happen in more than 1 in 10 people. There are things you can do to help you cope:
HeadachesMake sure you rest and drink plenty of fluids. Do not drink too much alcohol. Ask your pharmacist to recommend a painkiller.
Talk to your doctor if your headaches last longer than a week or are severe.
Feeling drowsy, sleepy or dizzyAs your body gets used to lamotrigine, these side effects should wear off. Do not drive, ride a bike or operate machinery until you feel more alert.
If they do not go within a week or two, your doctor may reduce your dose or increase it more slowly. If that does not work, speak to your doctor. You may need to switch to a different medicine.
Aggression, or feeling irritable or agitatedTalk to your doctor.
Shaking or tremorsTalk to your doctor if this is bothering you. These symptoms can be a sign that the dose is too high for you. It may help to change your dose or take your medicine at a different time of day.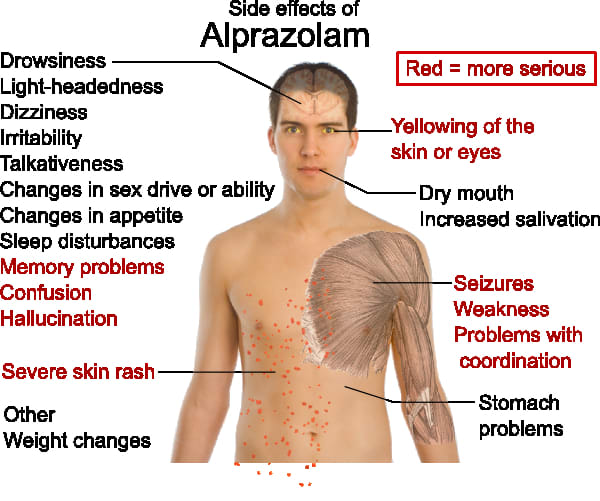
Talk to your doctor.
DiarrhoeaDrink lots of fluids, such as water or squash, to avoid dehydration. Signs of dehydration include peeing less than usual or having dark, strong-smelling pee.
Do not take any other medicines to treat diarrhoea without speaking to a pharmacist or doctor. Speak to a doctor if symptoms get worse or last longer than a week.
If you take contraceptive pills and you have severe diarrhoea for more than 24 hours, your contraception may not protect you from pregnancy. Check the pill packet to find out what to do.
Feeling or being sick (nausea or vomiting)Stick to simple meals and do not eat rich or spicy food. It might help to take your lamotrigine after a meal or snack. If you're being sick, take small, frequent sips of water or squash to avoid dehydration.
Speak to a doctor if symptoms get worse or last longer than a week.
If you take contraceptive pills and you're being sick, your contraception may not protect you from pregnancy. Check the pill packet to find out what to do.
Mild skin rashIf you get a mild rash speak to your doctor for advice as your treatment may need to be changed.
Also see our serious side effects information.
If this advice does not help and you are bothered by any of these side effects, keep taking the medicine but tell your doctor or pharmacist.
Serious side effects
Skin rashes
It's common to get a skin rash with lamotrigine. Most skin rashes are not serious.
Stevens-Johnson syndrome is a rare side effect of lamotrigine.
It causes flu-like symptoms, followed by a red or purple rash that spreads and forms blisters. The affected skin eventually dies and peels off.
The affected skin eventually dies and peels off.
It's more likely to happen in the first 8 weeks of starting lamotrigine, or when the dose is increased too quickly.
It can also happen if lamotrigine is stopped suddenly for a few days and then restarted at the same dose as before, without reducing the dose and then increasing it slowly again.
Stevens-Johnson syndrome is more common in:
- children
- people who have developed a rash before with a different epilepsy medicine
- people who are allergic to an antibiotic called trimethoprim
- people also taking a medicine called sodium valproate
To help reduce the chance of you getting a rash that could be confused with Stevens-Johnson syndrome, it's best to not try any new medicines or food during the first 3 months of treatment with lamotrigine.
Immediate action required: Go to A&E now if:
- you get a severe rash with flushing, blisters or ulcers – these can be signs of Stevens-Johnson syndrome
Find your nearest A&E
Other serious side effects
Very few people taking lamotrigine have any serious problems as any serious reaction is quite rare.
Tell a doctor or contact 111 straight away if you have a serious side effect, including:
- worsening fits or seizures (if you take lamotrigine for epilepsy)
- unexpected bruising or bleeding, a high temperature or sore throat – these could be signs of a blood disorder
Go to 111.nhs.uk or call 111.
Immediate action required: Go to A&E or call 999 now if:
- you have thoughts of harming or killing yourself – a small number of people taking lamotrigine for bipolar disorder have had suicidal thoughts, and this can happen after only a few weeks of treatment
- you have a stiff neck, headaches, feel or are being sick, a high temperature and extreme sensitivity to bright light – these could be signs of meningitis
Serious allergic reaction
In rare cases, it's possible to have a serious allergic reaction (anaphylaxis) to lamotrigine.
Immediate action required: Call 999 or go to A&E now if:
- you get a skin rash that may include itchy, red, swollen, blistered or peeling skin
- you're wheezing
- you get tightness in the chest or throat
- you have trouble breathing or talking
- your mouth, face, lips, tongue or throat start swelling
You could be having a serious allergic reaction and may need immediate treatment in hospital.
Long-term side effects
Long-term treatment with lamotrigine can cause osteoporosis and osteopenia, increasing your risk of breaking a bone.
Your doctor can arrange for tests to check your bone strength.
Regular exercise and a good diet can also help keep your bones strong.
Other side effects
These are not all the side effects of lamotrigine. For a full list, see the leaflet inside your medicine packet.
Information:
You can report any suspected side effect using the Yellow Card safety scheme.
Visit Yellow Card for further information.
Page last reviewed: 20 May 2022
Next review due: 25 May 2025
Lamotrigine side effects and safety information
Lamotrigine is a generic prescription drug used to prevent certain types of seizures as well as treat bipolar I disorder and migraines
Lamotrigine side effects | Serious side effects of lamotrigine | How long do side effects last? | Warnings | Interactions | How to avoid side effects
Lamotrigine is a generic prescription drug primarily used to prevent certain types of seizures in people with epilepsy (including Lennox-Gastaut syndrome) as well as to help stabilize moods in people with bipolar I disorder. For both types of conditions, lamotrigine is used alone or combined with other antiepileptic or bipolar medications.
For both types of conditions, lamotrigine is used alone or combined with other antiepileptic or bipolar medications.
Lamotrigine is sometimes used off-label to prevent migraines. Available also under the brand names Lamictal and Lamictal XR (an extended-release form of lamotrigine), lamotrigine works by slowing down nerve activity, making seizures and brain excitement less likely. Lamotrigine does have risks, however, so a careful review of side effects, drug interactions, restrictions, and other warnings will help people taking lamotrigine to manage these risks.
RELATED: Learn more about lamotrigine
Common side effects of lamotrigine
Because lamotrigine affects the nervous system, it can affect the entire body. The most common side effects of lamotrigine are:
- Dizziness
- Headache
- Double vision
- Loss of coordination
- Blurred vision
- Nausea
- Sleepiness or insomnia
- Weakness
- Nasal congestion and runny nose
- Sore throat
- Skin rash
- Vomiting
- Back or abdominal pain
- Fatigue
- Dry mouth
Serious side effects of lamotrigine
Lamotrigine can cause serious side effects such as:
- Seizure lasting longer than 5 minutes (status epilepticus)
- Withdrawal seizures if the medication is stopped abruptly
- Suicidal thoughts and behaviors
- Worsening of depression
- Aseptic meningitis (swelling of the membranes surrounding the brain)
- Liver failure
- Irregular heartbeat
- Blood problems
- Kidney problems
- Immune system problems such as hemophagocytic lymphohistiocytosis and vasculitis
- Severe or life-threatening hypersensitivity reactions including serious skin rash, Stevens-Johnson syndrome, angioedema, and toxic epidermal necrolysis
How long do side effects last?
Lamotrigine has a half-life of about 25 hours in healthy people, meaning it takes about 25 hours to clear half of the medication dose from the body.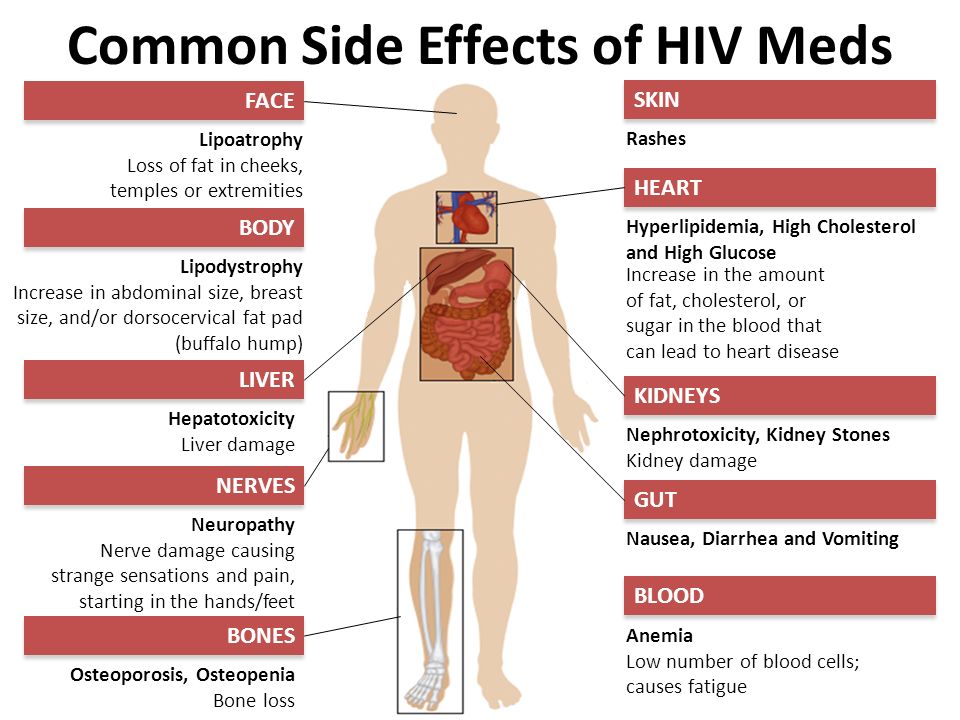 It takes about 4 to 5 half-lives to clear most of a drug from the body. Most temporary side effects, such as headache, dizziness, drowsiness, nausea, rash, and vision problems, then, will take about 4 to 5 days to fade after taking the last dose of lamotrigine. Some side effects may get better as the body adjusts to the medicine. The prescribing information states that lamotrigine should be discontinued at the first sign of rash unless the rash is definitely not related to the medication. This is because a rash caused by lamotrigine could be anywhere from mild to serious or life-threatening.
It takes about 4 to 5 half-lives to clear most of a drug from the body. Most temporary side effects, such as headache, dizziness, drowsiness, nausea, rash, and vision problems, then, will take about 4 to 5 days to fade after taking the last dose of lamotrigine. Some side effects may get better as the body adjusts to the medicine. The prescribing information states that lamotrigine should be discontinued at the first sign of rash unless the rash is definitely not related to the medication. This is because a rash caused by lamotrigine could be anywhere from mild to serious or life-threatening.
More severe side effects may require medical treatment to resolve, particularly meningitis, blood problems, immune system problems, severe allergic reactions, and status epilepticus. Many of these are life-threatening conditions that require immediate emergency medical care. Some conditions may have lifelong consequences.
Lamotrigine contraindications & warnings
Several red flags may prompt a healthcare provider to either avoid prescribing lamotrigine or doing so only cautiously.
Abuse and dependence
The U.S. Food and Drug Administration (FDA) does not classify lamotrigine as a controlled substance. Although it has sedative effects, it is rarely used recreationally because of its high rate of side effects.
Lamotrigine does not cause physical dependence, but sudden discontinuation of the drug may cause seizures even in patients taking lamotrigine for conditions other than epilepsy. When it’s time to stop lamotrigine, a healthcare provider will use a steadily tapering dose (over about 2 weeks) to prevent seizures.
Lamotrigine overdose
Depending on how much medicine is taken, an overdose of lamotrigine is a medical emergency and could be fatal. Symptoms of an overdose include:
- Impaired coordination
- Repetitive uncontrolled eye movements (nystagmus)
- Blurred vision
- Abnormal heartbeat
- Seizures and convulsions
- Reduced consciousness, fainting, or loss of consciousness
- Coma
If there’s any sign or suspicion that too much lamotrigine has been taken, seek emergency medical care.
Contraindications
Lamotrigine should never be taken by people who are allergic to the drug or any of its inactive ingredients. Any severe allergic or immunological reaction while taking lamotrigine will result in the drug being immediately discontinued.
Kidney or liver disease
People with significant renal or moderate to severe hepatic impairment may require a lower dose. People with mild liver impairment should be able to receive a normal dose. The prescribing information advises using lamotrigine with caution in patients with severe kidney impairment.
Heart problems
Because lamotrigine can change the heart’s rhythm, lamotrigine is used cautiously in people with heart failure, heart disease, heart rhythm problems, ischemic heart disease, heart conduction problems such as Brugada syndrome, ischemic heart disease, or who have risk factors such as coronary artery disease. Medically serious and potentially fatal changes in the heart’s rhythm are possible, so healthcare providers are very careful about using lamotrigine in these patients.
Depression
Lamotrigine can worsen depression and even provoke suicidality. Anyone taking lamotrigine, especially people with depression or a history of suicidality, will need to be monitored carefully by both healthcare professionals and caregivers for signs of increased depression, behavior changes, suicidal thoughts, and suicidal or risk-taking behaviors.
Pregnancy and breastfeeding
There is no evidence that taking lamotrigine during pregnancy will harm a fetus, but there is conflicting data between human and animal studies. Your healthcare provider can determine if lamotrigine is safe for you during pregnancy. Lamotrigine may decrease folate levels, so healthcare providers will suggest taking folic acid supplements.
Lamotrigine does pass into breast milk, so infants who are nursing may experience lamotrigine side effects such as rash, drowsiness, trouble breathing, reduced feeding, and poor weight gain. Infants may need to be monitored for side effects. Alternatively, nursing women may be asked to stop nursing while taking lamotrigine.
Alternatively, nursing women may be asked to stop nursing while taking lamotrigine.
Children
Lamotrigine is FDA-approved as an add-on drug in children as young as 2 years old in treating partial-onset seizures, generalized seizures of Lennox-Gastaut syndrome, and primary generalized tonic-clonic seizures. It is not approved as a single therapy or as a treatment for any other condition in children or teens. Children taking lamotrigine are more likely to experience infections than adults. In addition, children taking lamotrigine experience a high rate (14%) of accidental injury, though accidental injury is not a common side effect among adults.
Seniors
People aged 65 years and older can be prescribed lamotrigine, but healthcare providers will typically start older patients with a low dose and monitor them carefully for side effects.
Lamotrigine interactions
Several drugs can affect lamotrigine or increase the risk of side effects.
Contraindications
Some drugs are never combined with lamotrigine because they significantly raise the risk of seizures. These contraindicated drugs include amifampridine, dalfampridine, Wellbutrin (bupropion), and metoclopramide.
These contraindicated drugs include amifampridine, dalfampridine, Wellbutrin (bupropion), and metoclopramide.
Other drug interactions
Other drugs can be taken with lamotrigine, but the combination will be avoided if possible or the patient will need dosage adjustments or careful monitoring. These include:
- Drugs such as birth control pills, female hormone replacements, ritonavir, and rifampin.
- Anticonvulsant drugs such as phenobarbital, phenytoin, primidone, and carbamazepine may be prescribed along with lamotrigine but careful dosage adjustments are needed by healthcare providers.
- Antiepileptic drug Depakote (divalproex sodium). One or both medication dosages should be adjusted by a trained healthcare professional.
- Central nervous system depressants such as alcohol, opioids, barbiturates, benzodiazepines, sedatives, migraine medications, antihistamines, muscle relaxants, SSRIs, and MAO inhibitors.
 While these drugs don’t have to be avoided outright, increased drowsiness and impairment may significantly affect day-to-day life.
While these drugs don’t have to be avoided outright, increased drowsiness and impairment may significantly affect day-to-day life.
How to avoid lamotrigine side effects
Unfortunately, most people taking lamotrigine will experience one or more side effects. Many of these side effects are minor and will improve as the body adjusts to the drug. Still, there are ways to minimize side effects or increase the benefits of taking lamotrigine.
1. Tell the healthcare provider about all medical conditions and medications
The first step in preventing side effects is to tell the healthcare provider prescribing lamotrigine about all medical conditions, especially:
- Any history of a rash or skin reactions to other epilepsy medications
- Any history of depression, mood problems, or suicidality
- Any heart problems or heart rhythm problems
- Any history of aseptic meningitis while taking lamotrigine
- Kidney or liver disease
- Pregnancy or pregnancy plans
- Breastfeeding or plans to breastfeed
Some side effects are caused or worsened by combining lamotrigine with other prescription or over-the-counter drugs.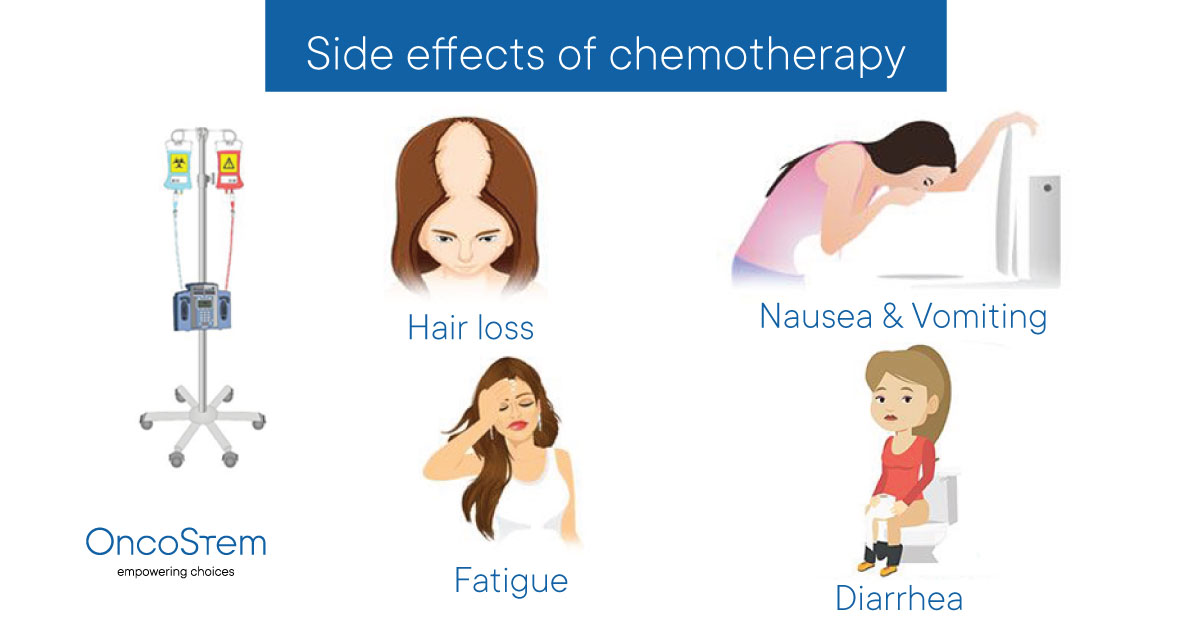 Keep a list of all medications being taken and share it with the healthcare provider prescribing lamotrigine. They will especially need to know about oral contraceptives or female hormone replacement therapy.
Keep a list of all medications being taken and share it with the healthcare provider prescribing lamotrigine. They will especially need to know about oral contraceptives or female hormone replacement therapy.
2. Take lamotrigine as directed
Take lamotrigine exactly as instructed. The dose will vary depending on the condition being treated, underlying medical conditions, and other medications prescribed. For lamotrigine to work, doses must be taken every day. Don’t skip a dose. If prescribed orally disintegrating tablets, chewable tablets, or dispersible tablets, follow the instructions carefully. They are not taken in the same way as tablets. If there are any questions about these instructions, consult with a pharmacist or other healthcare provider.
3. Avoid alcohol
Lamotrigine makes many people sleepy and can cause other impairments such as loss of coordination, double vision, or other problems. Drinking and lamotrigine don’t mix well. Alcohol will worsen the impairment caused by lamotrigine and put people at an increased risk of injury or accident.
4. Be careful about driving and other risky activities
In many people, lamotrigine can cause drowsiness and vision problems. Side effects like sleepiness and double vision can make simple daily activities such as driving or operating machinery hazardous. In many cases, these side effects will lessen as the body adjusts to lamotrigine. Avoid driving or other risky activities until lamotrigine’s impairment effects are well-understood or have faded.
5. Drink fluids
If diarrhea is experienced, drink plenty of fluids to avoid dehydration.
6. Talk to a healthcare provider
You should immediately call a healthcare provider if you notice side effects such a:
- Skin rash
- Blistering
- Hives
- Mouth or eyesores
- Fever
- Stiff neck
- Muscle pain
Some other side effects such as shaking or tremors may be a sign that the dose is too high. Talk to the doctor or other healthcare provider if you have these side effects or others such as sleep disruption or irritability. Dosing may need to be adjusted.
Talk to the doctor or other healthcare provider if you have these side effects or others such as sleep disruption or irritability. Dosing may need to be adjusted.
7. Watch for signs of heart problems
Lamotrigine can cause irregular heartbeats, so people taking lamotrigine should immediately contact a healthcare provider or seek emergency medical treatment at any sign of heart problems such as:
- Fast or slow heartbeat
- Chest pain
- Shortness of breath
- Lightheadedness
8. Do not stop taking lamotrigine
Going cold turkey with lamotrigine may cause seizures even if the drug is prescribed for another condition. If the drug does not seem to be working or, worse, the side effects are too hard to take, see a healthcare professional for medical advice. The safest way to stop taking lamotrigine is with a steadily tapering dose—over several weeks—supervised by a healthcare provider.
9. Inspect your medication each time you fill your prescription.
Although mixups are very rare, due to multiple formulations as well as other drugs that are sometimes confused with lamotrigine (due to look-alike or sound-alike drug names), it is a good idea to check your medication each time you fill your prescription. If the medication looks different than the previous refill, check with your pharmacist to make sure you received the correct drug.
Related resources for lamotrigine side effects:
- How to minimize lamotrigine’s adverse effects, Psychiatric Times
- Lamotrigine, Epocrates
- Lamotrigine compound summary, U.S. National Library of Medicine
- Lamictal prescribing information, U.S. National Library of Medicine
- Lamotrigine tablets prescribing information, U.S. National Library of Medicine
- Studies show increased risk of heart rhythm problems with lamotrigine in patients with heart disease, FDA
| Treatment of epilepsy involves long-term use of antiepileptic drugs (AEDs), but sometimes, after some time after the start of treatment, the previously prescribed AED has to be replaced with another one. Distinguish between acute and chronic toxic effects that antiepileptic drugs have [1]. Among them, non-specific dose-dependent, specific pharmacodynamic effects for a given drug and rare, but potentially life-threatening idiosyncratic reactions are distinguished. The first two types of reactions can usually be diagnosed and prevented in a timely manner. The latter type of reactions, as a rule, is unpredictable and develops very quickly, which practically does not allow diagnostics at the preclinical stage [2-5]. The main serious side effects of this type include hepatitis, pancreatitis, bone marrow suppression, and exfoliative dermatitis. These effects, if not recognized in time, can lead to the death of the patient [6], and the reaction does not depend on the dose of the drug and is unique in each case [1]. nine0007 Potentially life-threatening idiosyncratic reactions are listed in Table 1. Idiosyncratic reactions usually develop within the first 6 months after starting the drug. Their frequency is approximately 1:30,000 - 1:50,000. This risk is higher for the development of Stevens-Johnson syndrome for lamotrigine - about 1:1000 [7], aplastic anemia for felbamate - about 1:5000 [8] and liver damage in young children with polytherapy with the inclusion of valproic acid preparations - about 1:500 [9]. Hypersensitivity syndrome to AED is observed at 2-8 weeks of treatment with aromatic AED. Hypersensitivity syndrome to AED is observed at 2-8 weeks of treatment with aromatic AED. Symptoms are rash, fever, and eosinophilia. Lymphadenopathy and potentially life-threatening necrotic changes in the liver may also occur [10]. More severe reactions include exfoliative dermatitis, erythema multiforme, Stevens-Johnson syndrome, and toxic epidermal necrolysis [11], which is presumably based on the pharmacogenetic features of biotransformation [12]. When prescribing any AED that is at risk of developing Stevens-Johnson and Lyell syndrome or anticonvulsant hypersensitivity syndrome [13], the patient (parents or caregivers) should be warned of the risk of rash and the need for daily skin examination. The patient should be instructed on what action to take if a rash is detected. The doctor should plan in advance a possible reserve AED, which has two properties: no risk of developing skin complications and the possibility of prescribing a therapeutic dose from the first day (valproate, levetiracetam). nine0007 There are currently no reliable ways to prevent idiosyncratic reactions. Currently, there are no reliable ways to prevent idiosyncratic reactions. Taking into account the proposed mechanism of immediate type hypersensitivity [14], it is proposed to use a mast cell degranulation test before prescribing a drug or a lymphocyte transformation reaction, taking into account the alleged role of epoxide hydrolase deficiency (a component of the cytochrome oxidase system) in the pathogenesis of these conditions [12,15]. Neurotoxic effects in the form of drowsiness, dizziness, affective changes (depression, agitation), impaired memory, attention, performance when using AED are quite common [1,16]. Tracking these effects is especially necessary in the early stages of treatment, because. over time, the patient forgets his "normal" state - the one that was before the appointment of the drug. A few months after the appointment of AED, the patient himself and his relatives begin to perceive the "new" state as "normal". And only after discontinuation of the drug, patients often notice that the world around them began to be perceived brighter, the level of cognitive functioning increased, and working capacity increased. Thus, the so-called. the phenomenon of "adaptation to the drug" does not mean that the neurotoxic effect disappears over time, the patient simply forgets how he felt before treatment [4]. In the early stages of therapy, it is especially important to ask the patient about his well-being, to try to accurately identify neurotoxic symptoms (for example, drowsiness with carbamazepine or slow thinking with topiramate). When taking AEDs, more pronounced neurotoxic effects may also be observed: tremor, diplopia, sometimes dysarthria, ataxia [1,16]. Neurotoxic effects can be pharmacokinetic and pharmacodynamic [17]. Therefore, when neurotoxic effects appear, it is necessary to determine the level of the drug in the blood to exclude the pharmacokinetic mechanism of their occurrence. Protocols for monitoring the presence of chronic specific adverse effects of AED are currently not published [4]. Most authors recommend conducting a preliminary examination, during which a risk group is identified in relation to the development of undesirable effects of the use of AEDs [4,17]. This group consists of patients with changes in biochemical parameters, neurodegenerative diseases, concomitant somatic diseases, and a history of severe idiosyncratic reactions [17]. The observation plan for the timely detection of the occurrence of undesirable effects depends on the specific clinical situation (type and doses of the drug used, risk factors). Table 2 lists the major serious adverse effects of AEDs. Below are descriptions of the main side effects of AEDs presented on the Russian pharmaceutical market. Barbiturates commonly cause behavioral problems in children (hyperactivity, irritability) and drowsiness in adults. Attention disturbances, thought disorders, and depression may be both dose-dependent and independent of plasma concentrations of the drug in sensitive patients [18]. Dose-dependent neurotoxic effects include nystagmus, ataxia, dyskinesia, and disruption of sleep patterns. nine0007 Idiosyncratic reactions include allergic dermatitis, Stevens-Johnson syndrome, serum sickness (serum sickness, the term is used in the English literature and for non-protein drugs) and liver damage, agranulocytosis and aplastic anemia [5]. Exacerbation of porphyria may be noted [17]. Being inducers of microsomal enzymes, barbiturates alter the metabolism of vitamin D, which leads to the development of osteoporosis, especially in childhood. Being inducers of microsomal enzymes, barbiturates change the metabolism of vitamin D, which leads to the development of osteoporosis, especially in childhood and immobilized patients [17]. In addition, barbiturates impair absorption and accelerate the metabolism of vitamin K. In this regard, newborns from mothers receiving barbiturates need to be given vitamin K to prevent hemorrhagic disease of the newborn. The pregnant woman should receive vitamin K during the last month of pregnancy, at the beginning of the birth process, and the child should be given vitamin K immediately after delivery [17]. nine0007
Barbiturates interfere with absorption and accelerate the metabolism of vitamin K, therefore, newborns from mothers receiving barbiturates need vitamin K to prevent hemorrhagic disease of the newborn. With chronic use of barbiturates, changes in the connective tissue can be observed with the development of coarsening of facial features, Dupuytren's contracture, plantar fibromatosis and stiffness of the shoulder joint [17]. Phenytoin is characterized by non-linear pharmacokinetics and a narrow therapeutic range, which often leads to the development of dose-dependent adverse effects (nystagmus, ataxia, dyskinesia and cognitive impairment) not only at the beginning of treatment due to drug accumulation [5]. Children may sometimes be irritable or hyperactive. The combination of anorexia, vomiting and weight loss are characteristic of phenytoin intoxication in childhood [17]. There are also a number of dose-independent effects. With prolonged use of the drug, there is a coarsening of facial features, a change in the texture and color of the hair, gum hyperplasia, the appearance of acne, the development of osteoporosis and lymphadenopathy. Folic acid deficiency with phenytoin can lead to megaloblastic anemia and transient encephalopathy. nine0007 Folic acid deficiency with phenytoin can lead to megaloblastic anemia and transient encephalopathy. Long-term elevated plasma levels of phenytoin can lead to cerebellar atrophy. Potentially life-threatening idiosyncratic reactions include allergic skin reactions, hepatopathy, serum sickness-type reaction, and aplastic anemia [5]. The development of a systemic drug reaction in the form of lupus erythematosus has also been described [17]. nine0007 Ethosuximide in a high single dose causes vomiting, abdominal pain. These phenomena can be reduced by taking the drug during meals and by reducing the single dose by increasing the frequency of taking the drug. Rare idiosyncratic effects include skin rashes, leukopenia, pancytopenia, and aplastic anemia [10]. Neurotoxic effects include headaches, which can be very intense, as well as drowsiness, agitation, aggressiveness, depression, and memory impairment. Mental disorders are associated with the phenomenon of forced normalization of the EEG. In children, the development of a systemic drug reaction similar to lupus has also been described [17]. Carbamazepine causes a moderate dose-dependent neurotoxic effect. At the beginning of treatment, transient symptoms may occur in the form of nausea, drowsiness, dizziness, ataxia and blurred speech, which regress within a week without changing the dose of the drug. Diplopia, tremor and headache occur with an increase in plasma concentrations of the drug and can serve as a clinical marker during dose titration [16]. Occasionally, nausea, vomiting, diarrhea, and abdominal pain may occur. Hyponatremia is common but rarely reaches a clinically significant level. These dose-dependent effects are less pronounced when using prolonged forms of the drug [17]. nine0007 Skin rashes occur in 5-8% of patients, but exfoliative dermatitis and bullous-type reactions (eg, Stevens-Johnson syndrome) are rare [16]. Hematological changes occur quite often - leukopenia is noted in 10-12% of patients. Leukopenia occurs in 10-12% of patients receiving carbamazepines. However, fatal reactions such as aplastic anemia are rare, with a mortality rate of about 1.1:500,000 treated with carbamazepine per year. Despite the fact that this opinion is not shared by all authors [4,17], we consider it appropriate to conduct hematological control to exclude the progressive nature of leukopenia. As suggested by the Canadian Association of Child Neurologists [19], we perform a clinical blood test with platelet count before starting treatment. Then, taking into account the life span of cytopopulations, it is advisable to carry out hematological control after 5-6, 14-16 and 30 days from the start of taking the drug (or increasing its dose). In the future, in the first year of treatment, hematological control is carried out 1 time in 3 months, with a longer use of the drug - 1 time in 6 months. Taking into account the possible hepatotoxic effect [16], we suggest monitoring the activity of liver enzymes within 1 month from the start of taking the drug, then once every 6 months and according to indications. Valproates can cause gastrointestinal symptoms - nausea, vomiting, abdominal pain, diarrhea, which in most patients disappear when prolonged forms of the drug are prescribed and taken with food. Tremor during stress and at rest depends on the dose of the drug and, possibly, the age of the patient, because. in children, this symptom is less common. An increase in body weight due to an increase in appetite is observed in 20-54% of patients, which in some cases requires discontinuation of the drug. Hair loss is another common unwanted effect. The effect is transient. Hair becomes brittle, and newly growing hair changes its structure, becoming more curly. The use of multivitamins containing folic acid and zinc can reduce this undesirable phenomenon. There is evidence of the possibility of developing polycystic ovaries during treatment with valproate. There is evidence of the possibility of developing polycystic ovaries during treatment with valproate [21]. In this regard, it is recommended that girls and women undergo pelvic ultrasound with the determination of the volume of the ovaries before the start of treatment, followed by control once a year. Idiosyncratic reactions include acute encephalopathy and coma, which may occur early in treatment. In a laboratory study, pronounced acidosis and increased excretion of organic acids are revealed. It is assumed that this complication occurs in patients with a previously compensated defect in mitochondrial beta-oxidases [22,23]. Allergic skin reactions are extremely rare, but can be quite severe [24]. Acute hemorrhagic pancreatitis can be fatal, and therefore in patients with abdominal pain during treatment with valproate, it is necessary to determine the level of lipase and amylase [17]. nine0007 The most severe idiosyncratic reaction associated with this drug is fulminant irreversible liver damage. Most often, this complication occurs against the background of polytherapy in children under the age of 2 years, with severe epilepsy, mental retardation, and signs of organic brain damage [9]. Routine laboratory tests do not allow to identify a risk group and catch the initial signs of liver damage: in many patients with advanced liver damage, laboratory data remained within the normal range, while in other patients there is often a change in liver tests without evidence of clinically significant liver damage [23] . Gabapentin causes mild neurotoxic manifestations, but severe behavioral disorders (aggression, hyperactivity) may occur in mentally retarded children. Lamotrigine may cause neurotoxic effects such as fatigue, drowsiness and confusion [28]. A feature of the beginning of treatment with lamotrigine is the need for a slow dose titration. This is due to the risk of developing skin rashes in the first 8 weeks of therapy, in rare cases, up to the development of Stevens-Johnson and Lyell syndromes. nine0007 The risk of rash when lamotrigine 5-10% is used with valproate increases to 14%. The risk of developing a rash when lamotrigine 5-10% is used together with valproate increases to 14%. Predictors of the development of a rash are: a rash against the background of another AED in history; children under 13 years of age; combined use with valproate [29]. Currently, slow titration of the dose of lamotrigine is considered mandatory. Pregabalin may cause neurotoxic effects (dizziness, ataxia, drowsiness) and weight gain [30]. Levetiracetam may cause headache, irritability, drowsiness and anorexia [31]. Significant behavioral disorders and psychoses have been described in childhood [32]. Topiramate causes dose-dependent neurotoxic effects (ataxia, dizziness, paresthesia, impaired concentration, impaired thinking, fatigue, drowsiness, confusion, psychosis) [33]. Slow titration of the drug reduces the risk of these effects. Other serious complications in the treatment of this drug are weight loss, skin rashes. Acute secondary glaucoma may also occur, and therefore the patient should be warned about the need to immediately contact a specialist in case of pain in the eyes and visual impairment [34]. Oxcarbazepine is a derivative of carbamazepine, which is rapidly converted to the active metabolite 10,11-dihydro-10-hydroxycarbamazepine, due to which its toxic effects are less pronounced compared to carbamazepine. Dose-dependent neurotoxic effects (dizziness, drowsiness, fatigue) may occur. Hyponatremia can be quite pronounced. Skin rashes, Stevens-Johnson syndrome, and exfoliative dermatitis are rare with oxcarbazepine [36]. Thus, the prescription of any AED carries the risk of side effects. It is necessary to identify groups of patients where this risk may be increased. The patient (or his parents, guardian) must be warned about the occurrence of possible undesirable effects. During the treatment of epilepsy, it is necessary to carry out clinical and laboratory monitoring according to an individually drawn up observation plan for the earliest possible detection of possible side effects, especially in the first six months of treatment with the drug. If signs of undesirable effects appear, the patient should contact the attending physician to resolve the issue of correcting antiepileptic therapy. nine0007 Table 1. Serious side effects of AEP (according to Schmidt D. [13])
How to take antiepileptic drugs (AEDs)home Services School of epilepsy How to take antiepileptic drugs (AEDs) GeneralAntiepileptic drugs (AEP) reduce the excessive excitability of brain cells, preventing the development of epileptic seizures. The effect of AEDs depends on their concentration in the blood, so the drugs should be taken daily. Most modern AEDs must be taken twice a day (morning and evening) to keep their blood levels stable. nine0007 How to increase the dose of the drug? The slower the dose is increased, the lower the risk of side effects. Usually start with ½ or 1 tablet in the evening, after a few days add ½ or 1 tablet in the morning and so on, bringing the dose to therapeutic. What if I miss a dose?For such cases, drugs have been developed that must be taken 2 times a day. If you forgot to drink your morning dose and remembered about it a few hours later, feel free to take it. If you remembered the forgotten dose only in the evening, you should not drink the morning + evening dose. Drink only the evening drink and do not forget to take the drug again. nine0007 How do I remember to take my medication?It is necessary to "attach" the drug intake to regular events in life, for example: after breakfast and after dinner, after morning and evening walks with the dog, after morning and evening washing. You can use reminders on your mobile phone. If the patient suffers from impaired memory and thinking, relatives should monitor the medication. We recommend that you keep a diary of seizures every day and record each medication in it. How to take medicines: before or after meals? For modern drugs, this is not important. How do I change from one drug to another?The most important rule is to do everything gradually. In most cases, the dose of the "new" drug is gradually increased first, then the dose of the "old" drug is also gradually reduced. How to replace the drug with its analogue from another manufacturer (after all, the active substance is the same there)? nine0542The universal rule is that if the body is accustomed to a certain drug of a certain company, then it is better not to change it. However, if there is no other choice (they stopped giving the drug for free and offer its substitute, but there is no money to buy the old drug), then it is necessary to gradually replace the “old” pills with the “new” ones. If you abruptly quit the “old” ones and start drinking the “new” ones, the risk of developing seizures and side effects is higher. How dangerous are AEDs to health? Unfortunately, absolutely safe drugs do not exist. A) Idiosyncratic reactions (individual intolerance). They develop in the first months from the start of the medication. Most often - skin rash, jaundice, severe abdominal pain, bleeding, severe vomiting (there may be other manifestations). Idiosyncrasy appears suddenly, it cannot be predicted in advance. At risk are patients who have previously had severe adverse reactions to any drugs. If after the start of taking the medicine there are any serious disorders, you should immediately stop taking it and consult a doctor. nine0007 B) Dose-dependent side effects (they are not on small doses, but they appear on large ones). Typical examples: drowsiness, double vision, unsteadiness when walking, lethargy, trembling in the hands, etc. |
 One of the important reasons for the forced replacement of the drug is its side effects.
One of the important reasons for the forced replacement of the drug is its side effects. 
 nine0007
nine0007  nine0007
nine0007  Comparison of the results of simple non-labor-intensive tests to assess the level of attention, working capacity, short-term memory and psychomotor tests before treatment and in the first months after its start reveals the appearance of neurotoxic effects of the drug. We suggest conducting such testing before prescribing the drug, and then, in the absence of complaints, after 1 month from the start of taking the drug and then 1 time in 3 months in the first year of treatment and 1 time in 6 months thereafter. In children, it is advisable not to compare individual indicators during treatment in "raw" scores, but to conduct a centile assessment of test performance, because standards change with age. nine0007
Comparison of the results of simple non-labor-intensive tests to assess the level of attention, working capacity, short-term memory and psychomotor tests before treatment and in the first months after its start reveals the appearance of neurotoxic effects of the drug. We suggest conducting such testing before prescribing the drug, and then, in the absence of complaints, after 1 month from the start of taking the drug and then 1 time in 3 months in the first year of treatment and 1 time in 6 months thereafter. In children, it is advisable not to compare individual indicators during treatment in "raw" scores, but to conduct a centile assessment of test performance, because standards change with age. nine0007  With a high content of the drug in the blood, it is enough to reduce its dose, which will eliminate the side effect of the treatment. However, the presence of a dose-independent neurotoxic (pharmacodynamic) effect, as a rule, requires discontinuation of the drug. nine0007
With a high content of the drug in the blood, it is enough to reduce its dose, which will eliminate the side effect of the treatment. However, the presence of a dose-independent neurotoxic (pharmacodynamic) effect, as a rule, requires discontinuation of the drug. nine0007  nine0007
nine0007  nine0007
nine0007 

 nine0007
nine0007 
 Since the drug can lead to the development of atrioventricular blockade [16], it is desirable to monitor the ECG (1 month after the start of the drug or increasing its dose, in the future - 1 time in 6 months). nine0007
Since the drug can lead to the development of atrioventricular blockade [16], it is desirable to monitor the ECG (1 month after the start of the drug or increasing its dose, in the future - 1 time in 6 months). nine0007  The development of thrombocytopenia, which usually does not reach a clinically significant level, depends on the dose of the drug and requires a dose reduction, and in the presence of clinical symptoms (appearance of petechiae, ecchymosis) - and its cancellation [17, 20]. For timely diagnosis of thrombocytopenia, we determine the number of platelets after 14-16 and 30 days from the start of taking the drug (or increasing its dose). In the future, in the first year of treatment, control is carried out 1 time in 3 months, with a longer use of the drug - 1 time in 6 months. nine0007
The development of thrombocytopenia, which usually does not reach a clinically significant level, depends on the dose of the drug and requires a dose reduction, and in the presence of clinical symptoms (appearance of petechiae, ecchymosis) - and its cancellation [17, 20]. For timely diagnosis of thrombocytopenia, we determine the number of platelets after 14-16 and 30 days from the start of taking the drug (or increasing its dose). In the future, in the first year of treatment, control is carried out 1 time in 3 months, with a longer use of the drug - 1 time in 6 months. nine0007 
 The combination of nausea, vomiting and anorexia was observed in 82% of cases of fulminant hepatopathy, and drowsiness, decreased level of consciousness and coma - in 40% [9]. Most often, this complication develops in the first 6 months of treatment, but can also be observed within 2 years after taking valproates [9]. Specific disorders associated with an increased risk of developing acute hepatopathy include: urea cycle disorders, organic aciduria, multiple carboxylase deficiencies, mitochondrial or respiratory chain dysfunction, cytochrome aa3 deficiency in muscle tissue, pyruvate carboxylase deficiency, and insufficiency of the hepatic pyruvate dehydrogenase complex [25 ], gangliosidosis type 2 GM1, spinocerebellar degeneration, Friedreich's ataxia, Lafort's disease, Alper's disease, MERRF syndrome [26]. nine0007
The combination of nausea, vomiting and anorexia was observed in 82% of cases of fulminant hepatopathy, and drowsiness, decreased level of consciousness and coma - in 40% [9]. Most often, this complication develops in the first 6 months of treatment, but can also be observed within 2 years after taking valproates [9]. Specific disorders associated with an increased risk of developing acute hepatopathy include: urea cycle disorders, organic aciduria, multiple carboxylase deficiencies, mitochondrial or respiratory chain dysfunction, cytochrome aa3 deficiency in muscle tissue, pyruvate carboxylase deficiency, and insufficiency of the hepatic pyruvate dehydrogenase complex [25 ], gangliosidosis type 2 GM1, spinocerebellar degeneration, Friedreich's ataxia, Lafort's disease, Alper's disease, MERRF syndrome [26]. nine0007  In addition, there may be an increase in body weight and peripheral edema with a normal content of protein and albumin in the blood plasma [27].
In addition, there may be an increase in body weight and peripheral edema with a normal content of protein and albumin in the blood plasma [27].  Strict adherence to this rule reduces the risk of developing severe, life-threatening rashes without reducing the overall risk of developing benign rashes. nine0007
Strict adherence to this rule reduces the risk of developing severe, life-threatening rashes without reducing the overall risk of developing benign rashes. nine0007  Another serious adverse effect associated with carbonic anhydrase inhibition is nephrolithiasis, which occurs in 1% of patients receiving topiramate [21], which requires monitoring of urine tests and periodic renal ultrasound for the detection of calculi. Increased fluid intake can reduce the risk of this complication. Children may develop hypohidrosis and hyperthermia; which is especially important in hot weather and during outdoor games. Parents need to be informed about this, because. a child with this complication needs additional cooling due to impaired heat transfer by convection [35]. nine0007
Another serious adverse effect associated with carbonic anhydrase inhibition is nephrolithiasis, which occurs in 1% of patients receiving topiramate [21], which requires monitoring of urine tests and periodic renal ultrasound for the detection of calculi. Increased fluid intake can reduce the risk of this complication. Children may develop hypohidrosis and hyperthermia; which is especially important in hot weather and during outdoor games. Parents need to be informed about this, because. a child with this complication needs additional cooling due to impaired heat transfer by convection [35]. nine0007  nine0007
nine0007 
 R. BOLDYREVA, A.Yu. .Sci., Children's City Hospital No. 1, St. Petersburg, Federal State Institution "Moscow Research Institute of Pediatrics and Pediatric Surgery of Rosmedtekhnologii"
R. BOLDYREVA, A.Yu. .Sci., Children's City Hospital No. 1, St. Petersburg, Federal State Institution "Moscow Research Institute of Pediatrics and Pediatric Surgery of Rosmedtekhnologii" 
 However, some of the AEDs can irritate the stomach, so we generally recommend taking it after a meal.
However, some of the AEDs can irritate the stomach, so we generally recommend taking it after a meal.  In the world, 80% of patients taking AEDs report some side effects. The more modern the drug, the more side effects are indicated in its instructions for use (the manufacturer indicates them so that it cannot be sued). Side effects can be divided into 3 groups: nine0007
In the world, 80% of patients taking AEDs report some side effects. The more modern the drug, the more side effects are indicated in its instructions for use (the manufacturer indicates them so that it cannot be sued). Side effects can be divided into 3 groups: nine0007